Diet-Responsive Chronic GI Disease in Dogs
Compound evidence detail2 SCRs / 5 parts
- Documentedcanine digestive anatomy and physiology evidence covering GI organs, AMY2B-linked starch adaptation, gastric acidity, transit-range variability, and microbiome fermentation
- Documentedthe bounded comparison to humans and wolves, where canine pH, transit, and starch-digestion findings support difference-from-wolf framing without diet absolutism
- Documentedthe AAFCO veterinary-exclusionary-statement pathway, the not-a-drug legal position of therapeutic diets, veterinary oversight boundary, and board-certified nutrition specialty framework
- Observed-JBcondition-category evidence where therapeutic diets have measurable support in areas such as renal disease, pancreatitis management, hepatic, urinary, or chronic-enteropathy care
- Heuristicthe category-wide efficacy boundary requiring condition-specific SCRs rather than universal claims across all therapeutic diet products
Chronic gastrointestinal disease is one of the most frustrating categories in family-dog medicine because the signs are messy, the causes overlap, and the internet encourages families to treat every soft stool as a referendum on one ingredient. Real chronic GI cases are usually more structured than that. Many dogs with vomiting, diarrhea, weight loss, or poor stool quality do improve dramatically with dietary change alone, which is why food-responsive disease is such an important category. But a chronic GI dog is not automatically a "sensitive stomach dog," and diet only works well when it is used inside an orderly diagnostic process. Documented
What It Means
The broad umbrella term many internists use is chronic enteropathy. This usually refers to gastrointestinal disease with signs such as chronic diarrhea, chronic vomiting, weight loss, reduced appetite, or mixed GI instability lasting weeks rather than days. Within that umbrella, clinicians often sort dogs by treatment response: food-responsive enteropathy, antibiotic-responsive enteropathy, steroid-responsive enteropathy, and non-responsive enteropathy. Observed-JB
That framework matters because it shifts the conversation away from guessing one diagnosis from the outside. Many chronic GI dogs are classified retrospectively by what actually works. A dog that normalizes on an appropriate diet trial fits the food-responsive bucket. Documented A dog that does not respond may move deeper into the workup. That is not diagnostic weakness. It is the reality of a disease group in which mechanism and treatment response are often closely linked.
Why Diet Comes First So Often
Food-responsive disease matters because it is common enough to justify trying it early in stable cases. Studies such as Allenspach and Mandigers helped establish that a substantial fraction of dogs with chronic GI signs improve or fully resolve on an appropriate dietary intervention alone. Documented That is a powerful finding because it means some dogs can avoid prolonged symptom cycles, repeated medication changes, or deeper invasive workups if the first diet step is chosen well and executed cleanly.
The key words there are chosen well and executed cleanly. A diet trial is not the same thing as grabbing any bag with "sensitive stomach" on the front. Depending on the case, the diet may be hydrolyzed, truly novel protein, highly digestible, low fat, or higher fiber. The target depends on the pattern of disease. A dog suspected of food-responsive enteropathy may need antigen simplification through hydrolysis or real novelty. A dog with lymphangiectasia or chronic pancreatitis crossover may need fat restriction. A dog with large-bowel signs may benefit from a different fiber strategy than a dog with small-bowel malabsorption.
The Main Clinical Categories
Food-responsive enteropathy is the category families most often hear about because it is the hopeful one. These dogs improve with diet alone or primarily with diet. The mechanism may involve immune reaction to dietary proteins, abnormal barrier interaction, altered microbial ecology, or a mix of these. The practical point is that the bowel calms when the right nutritional burden is removed.
Antibiotic-responsive enteropathy is the category many families know less well, partly because the profession has become more cautious about giving antibiotics casually. This is a good development. Years ago, chronic diarrhea dogs were often put on metronidazole early and repeatedly. Observed-JB Current practice is more guarded because antibiotics can muddy interpretation, damage microbial stability, and create their own problems.
Steroid-responsive enteropathy and non-responsive enteropathy occupy the more serious end of the spectrum. These are the dogs in whom diet alone is insufficient or only partially helpful and where inflammatory bowel disease, immune dysregulation, protein-losing enteropathy, or other deeper pathology becomes more likely. Families need this structure because it stops them from assuming every GI dog either needs lifelong prescription food or, alternatively, can be fixed with one boutique bag.
What Diets Actually Get Used
Hydrolyzed diets are commonly used when food-responsive disease is high on the list because they minimize intact protein exposure. Novel protein diets can also work, especially when the dog has not been heavily exposed to the chosen protein historically. Highly digestible diets are useful when the bowel needs a lower-residue, easier-to-handle nutrient stream. Low-fat diets matter in dogs where fat worsens signs or lymphatic disease is suspected. Documented Higher-fiber approaches matter more in selected large-bowel presentations.
This is why GI diet language can become confusing. Families hear one dog did well on fiber and another did poorly. One dog thrived on hydrolyzed protein and another needed low fat. Both stories can be true because "chronic GI disease" is not a single nutritional situation. It is a response-guided category.
The Microbiome Question
Families now hear a great deal about the microbiome, and some of that attention is justified. Chronic enteropathy is associated with measurable microbiome disruption, altered fermentation, and different metabolite patterns. But the microbiome should be handled carefully in family-facing writing. It is part of the explanation, not yet the full instruction manual. Saying chronic GI disease involves the microbiome is documented. Saying one specific supplement or feeding trend will normalize the microbiome and cure the disease usually outruns the evidence.
That is why the page's practical order remains conventional and disciplined: rule out obvious parasites and infectious causes, characterize the sign pattern, choose the right diet trial, monitor response, and escalate only when the response pattern demands it.
Why It Matters for Your Dog
This page matters because GI disease wears families down. Loose stool every week, vomiting every few days, waxing and waning appetite, middle-of-the-night urgency, and the constant fear that a new food will make everything worse turn daily care into a guessing game. When that uncertainty drags on, families either start changing diets too often or they become scared to change anything at all. Neither pattern helps.
For your dog, the biggest practical lesson is that many chronic GI cases are diet-responsive enough to justify taking the nutritional step seriously. That is hopeful. It means food is not just background support in these dogs. It can be real treatment. But it also means the food has to be selected for the disease pattern, not for marketing appeal.
The preventive move in chronic GI disease is not random trial-and-error feeding. It is a structured workup that gives the bowel one clean nutritional experiment at a time so the dog is not asked to live in endless digestive instability.
This page also matters because it keeps families from oversimplifying chronic GI disease into food allergy alone. Some food-responsive dogs are reacting to antigen exposure. Others may be responding to digestibility, fat restriction, fiber profile, or broader inflammatory quieting. A dog that improves on a hydrolyzed diet is not always proving the same thing as a dog that improves on a low-fat GI diet. The common denominator is that diet changed the disease trajectory.
Another practical reason this matters is that diet trials only work if the family understands the hidden failure points. Extra treats, flavored preventives, table food, sudden topper changes, or switching early because stool is not perfect by day five can all ruin interpretation. GI dogs often improve in stages. Some stabilize quickly. Others need a few weeks. Families who expect instant perfection often abandon a workable plan too early.
There is also a financial and emotional efficiency issue. A clean diet trial is often cheaper, calmer, and less invasive than months of fragmented retail experimentation. Even when the dog ultimately needs imaging, endoscopy, or immunosuppressive therapy, a disciplined initial diet step usually clarifies the path rather than wasting time.
It also helps families to know what a thoughtful initial workup usually includes before everyone jumps straight to lifelong prescription feeding. Stable chronic cases often still warrant fecal testing, giardia consideration, history review for dietary indiscretion, body-condition assessment, and basic laboratory screening. Depending on the dog, pancreatitis testing, cobalamin and folate assessment, abdominal imaging, or deeper intestinal workup may follow. The point is not to over-medicalize every soft-stool dog. It is to avoid the opposite mistake of assuming that weeks of GI signs are automatically solved by buying whichever sensitive-stomach bag has the strongest marketing.
The trial itself also deserves a more realistic timeline. Many families expect the stool to be perfect almost immediately or assume the food has failed if the first few days remain messy. Chronic enteropathy rarely behaves that neatly. Some dogs improve within a week, but others need several weeks for inflammation to settle, stool to normalize, and appetite confidence to return. That is one reason veterinarians often set a defined time frame in advance. The family is less likely to abandon the plan impulsively if everyone agreed from the beginning that the diet would be judged after an actual trial period rather than after a single disappointing bowel movement.
Another useful distinction is between control and cure. A dog that becomes normal on a hydrolyzed or novel-protein diet has gained something very important, but the family still needs to know whether that diet is the long-term maintenance answer, a diagnostic step before challenge, or a bridge into a broader management plan. Some GI dogs live comfortably for years on the first therapeutic diet that works. Others require later refinement because cost, palatability, protein source, or recurrence pushes the case into a more individualized plan.
It is also worth naming protein-losing enteropathy and lymphangiectasia, because families with chronic GI dogs sometimes encounter these terms late and suddenly. These are more serious manifestations in which nutrient loss, especially protein loss, becomes clinically important and low-fat strategy may take on much more urgency. The reason to mention them here is not to make every family fearful. It is to show why persistent weight loss, low albumin, edema, or severe chronic GI instability should move the case beyond family experimentation quickly.
The microbiome conversation becomes more helpful once it is placed under this broader logic. Dysbiosis is not a decorative buzzword. It tells us that chronic GI disease alters the intestinal ecosystem in meaningful ways. But the clinically mature use of that information is not to chase every new powder or fermented narrative. It is to recognize that diet choice, trial discipline, antibiotic restraint, and disease staging all influence that ecosystem. In other words, the microbiome belongs inside the structured workflow, not instead of it.
Families also benefit from tracking more than vague impressions during a diet trial. Stool frequency, stool form, vomiting episodes, appetite, weight trend, and any blood or mucus should be written down rather than remembered emotionally. That simple habit often reveals that a dog is improving more steadily than the household realized, or that the dog is not actually responding and needs escalation. Chronic GI disease is easier to manage when the family records patterns instead of relying on the memory of a stressful week.
That simple tracking habit also improves the veterinarian's next decision. Instead of hearing "he was better, then worse, then kind of the same," the clinician can see whether vomiting stopped first, whether stool quality improved gradually, whether weight stabilized, and whether a low-fat or hydrolyzed approach is actually earning its place. In chronic GI work, better observation is often part of better treatment.
When to See a Veterinarian
Veterinary evaluation is appropriate when vomiting or diarrhea becomes chronic, when soft stool keeps recurring after brief improvement, when weight loss is present, when appetite declines, or when blood or mucus repeatedly appears in stool. A chronic GI dog should not be managed indefinitely by label swapping alone.
Prompt evaluation matters even more if the dog is losing weight, has poor body condition, shows lethargy, has repeated vomiting, or seems painful after eating. Those signs raise the stakes and broaden the differential list beyond simple food responsiveness. Parasites, giardia, pancreatitis, protein-losing enteropathy, endocrine disease, foreign material, and chronic inflammatory disease all belong in the workup depending on the case.
Urgent evaluation is warranted for persistent vomiting, collapse, marked abdominal pain, black stool, large-volume bloody diarrhea, dehydration, or refusal to eat. Those are not diet-trial symptoms. They are urgent medical signs.
Families should also seek veterinary input if a dog improves partially on one diet but not fully. Partial response can be useful information, but it often means the case needs refinement rather than endless repetition of the same half-effective plan.
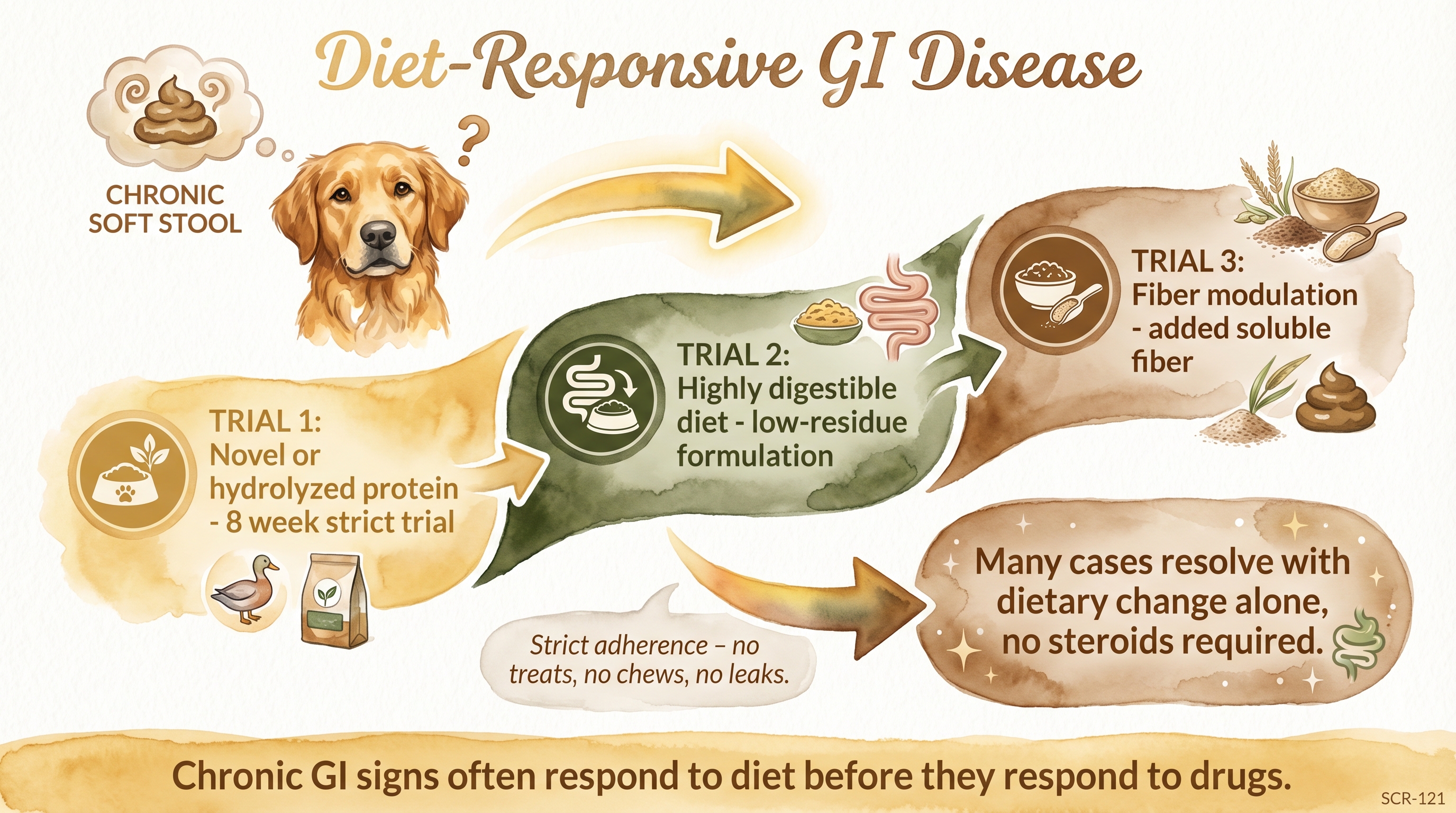
Chronic GI signs often respond to diet before they respond to drugs.
Key Takeaways
- Many dogs with chronic GI signs are genuinely diet-responsive, which is why a proper diet trial is often the first major treatment step in stable cases.
- Chronic enteropathy is a response-guided category, so hydrolyzed, novel-protein, low-fat, highly digestible, and fiber-modified diets each fit different patterns.
- A chronic GI dog should not be managed by random bag-switching because poor trial discipline makes the response impossible to interpret.
- Weight loss, repeated vomiting, blood in stool, or poor overall condition should push the case into veterinary workup rather than owner experimentation.
The Evidence
This entry uses observed claim-level tags beyond the dedicated EvidenceBlocks below. These tags mark JB program observation or practice-derived claims that need dedicated EvidenceBlock coverage in a later content pass.
The strongest evidence here is that a meaningful subset of dogs with chronic GI disease are food-responsive and can improve substantially on appropriately chosen dietary intervention alone. That is the core clinical takeaway. The next strongest point is that not every GI dog needs the same dietary strategy, which is why the category is response-guided rather than label-guided.
The evidence also supports keeping the microbiome in view without letting it become magical. Chronic enteropathy and dysbiosis are connected, but the family-facing instruction set is still led by clinical workflow more than by microbiome hype.
- Allenspach and Mandigers response-pattern literaturedogs with chronic enteropathy
A substantial fraction of dogs with chronic GI signs are food-responsive and may normalize on an appropriate dietary trial alone. - Clinical chronic-enteropathy frameworkdogs
Food-responsive, antibiotic-responsive, steroid-responsive, and non-responsive enteropathy remain useful practical categories because response pattern guides diagnosis and treatment. - Therapeutic diet evidence basedogs
Hydrolyzed, novel-protein, highly digestible, low-fat, and selected fiber-modified diets each have roles depending on disease presentation rather than one GI formula fitting every dog.
- Microbiome and chronic GI literaturedogs
Chronic enteropathy is associated with measurable microbiome disruption, but the practical management sequence is still driven by structured diagnostics and response-guided therapy rather than microbiome marketing. - Clinical workup synthesisdogs
Parasites, giardia, pancreatitis, and other medical causes must be considered before a family assumes the dog simply needs a different food forever.
No published study directly tests the practical implications of diet-responsive chronic gi disease in dogs for domestic dog raising programs. The application to household dog raising remains an interpretive synthesis rather than a directly tested intervention finding.
SCR References
Sources
-
National Research Council. (2006). Nutrient Requirements of Dogs and Cats. Washington, DC: The National Academies Press.
-
AAFCO. (2024). Official Publication. Association of American Feed Control Officials.
-
Allenspach, K., Wieland, B., Grone, A., & Gaschen, F. (2007). Chronic enteropathies in dogs: evaluation of risk factors for negative outcome. Journal of Veterinary Internal Medicine, 21(4), 700-708.
-
Mandigers, P. J., Biourge, V., & German, A. J. (2010). Foods and additives are common causes of adverse food reactions in dogs and cats. Journal of Nutrition, 130(12S), 1995S-1997S.