The HPA Axis in Dogs
Compound evidence detail2 SCRs / 4 parts
- Documentedthe canine methylation pattern reported by Awalt 2024 for NR3C1 and OXTR in dogs with measured early-life history
- Heuristicthe JB extrapolation that ordinary calm raising produces durable epigenetic advantages in dogs beyond preventing adverse environments
- Documentedthe Awalt 2024 finding that NR3C1 and OXTR methylation in dogs covaries with documented early-life experience
- Heuristicthe JB inference that those documented methylation differences in dogs predict specific later health or developmental outcomes
The hypothalamic-pituitary-adrenal axis, usually shortened to the HPA axis, is the core endocrine stress-response system in dogs. When a dog encounters novelty, separation, restraint, conflict, pain, transport, or other meaningful challenge, the HPA axis helps mobilize energy, shift attention, and coordinate physiological adaptation. In dogs, that architecture is well documented. The harder questions are how quickly it recovers, how early experience calibrates it, and what happens when activation becomes frequent enough that recovery starts to fail. Documented
What It Means
The Basic Architecture
The sequence is straightforward in outline. The hypothalamus releases corticotropin-releasing hormone (CRH), the pituitary releases adrenocorticotropic hormone (ACTH), and the adrenal cortex releases cortisol. Documented
Cortisol is the main glucocorticoid stress hormone in dogs. It helps mobilize fuel, alter immune allocation, and prepare the body to meet challenge. None of that is inherently pathological. In normal circumstances, the system is meant to rise, do work, and then stand down.
That recovery depends on negative feedback. Glucocorticoid receptors detect cortisol and help suppress further HPA output once the challenge has passed. Documented This is why stress physiology is not only about activation. It is also about shutoff. A resilient stress system is one that can respond and then recover.
Acute Activation Is Normal
One reason stress language becomes misleading so easily is that the HPA axis is often described as though any activation were harmful. That is not what the literature shows. Acute activation is adaptive. Handling, mild novelty, training challenge, separation, transport, and unfamiliar environments can all raise cortisol in dogs. That alone does not tell us the dog is being damaged.
The real question is whether activation is time-limited and followed by recovery, or whether the dog is repeatedly reactivated before the system has settled. This is the logic behind the acute-versus-chronic distinction that appears throughout modern stress science.
What Changes the System Over Time
The HPA axis is plastic, especially early in life. That is one reason this topic belongs under behavioral science rather than only under welfare. Stress physiology is part of development.
The classic epigenetic foundation comes from rodent maternal-care work. SCR-011 captures the finding that differences in maternal care alter methylation at glucocorticoid-related pathways and change later stress reactivity. Documented
For dogs, the key extension is Awalt et al. (2024), now reflected in SCR-094. Dogs with adverse early histories showed altered methylation patterns involving NR3C1 and OXTR together with differences in cortisol reactivity and attachment-related outcomes. That does not prove a deterministic life script. It does support a documented claim that early life can leave measurable marks on the machinery of stress regulation.
This distinction matters. The safe scientific statement is not "calm raising permanently programs a perfect dog." It is that early experience is biologically relevant to later HPA function, and dogs now have direct evidence for that claim.
Why It Matters for Your Dog
Recovery Matters as Much as Reactivity
If activation is only half the story, recovery is the other half. Stress systems become costly when stressors occur too frequently, the environment stays unpredictable long enough that shutoff is delayed, baseline arousal never falls fully between events, or later stressors arrive while earlier endocrine effects are still resolving.
That is why transition research matters so much. SCR-060 documents that moving dogs into novel kennel, shelter, or rehoming contexts produces marked cortisol disruption, and that quieter, more stable home environments help bring those measures down over time. Documented
The HPA axis therefore responds not only to isolated events, but to ecological context. A move, a new housing setup, social instability, or repeated uncontrollable challenge can become a stress landscape rather than a stress moment.
Downstream Effects of Dysregulation
Once the HPA axis is discussed over longer timescales, the conversation widens beyond cortisol alone. Chronic dysregulation can affect immune function, behavior and coping flexibility, mucosal defenses, cellular wear and tear, and attachment-related stress buffering. Documented
SCR-045 captures one of the most important downstream domains: chronic environmental stress in dogs alters immune-related outcomes, including lymphocyte and other cellular markers. Shelter, restriction, and adverse housing studies show that chronic stress is not just a feeling. It is accompanied by measurable physiological change.
This is also where caution is needed. The strongest direct canine evidence comes from sheltering, restriction, transport, and other adverse-environment paradigms. That does not license silent substitution of every household excitement pattern into the same category. The scientific principle is strong, but the stressor type still matters.
Why the HPA Axis Sits Under Calmness
The Five Pillars use calmness as a developmental principle. This page stays at the science layer, but the connection is clear enough to name: a dog's regulatory baseline depends in part on how often the HPA axis is asked to engage, how effectively it shuts off, and whether the caregiving environment helps or hinders recovery.
That does not mean calmness equals "no stimulation." It means that chronic activation without sufficient recovery has measurable costs, while stable environments and secure relationships can reduce those costs. The HPA axis is one of the reasons that claim is biological rather than purely philosophical.
The pillar page argues for building the calm floor first. The science page makes the narrower point: dogs possess a conserved mammalian HPA stress system, early life affects how that system is calibrated, and chronic dysregulation carries real physiological costs.
Key Boundaries
Several boundaries are worth stating explicitly. First, cortisol is not a synonym for damage. Acute rises can be adaptive. Second, negative feedback is central. The important question is not only how high the system goes, but how well it comes back down. Third, early-life programming language needs discipline. Rodent maternal-care epigenetics are foundational, but dog-specific claims should rely on the direct canine evidence now available rather than pretending the entire rodent mechanism has been mapped identically in dogs. Fourth, downstream immune and welfare claims are strongest in documented adverse-environment contexts. They should not be casually rewritten as proof that every excited or stimulating home is biologically equivalent to shelter stress.
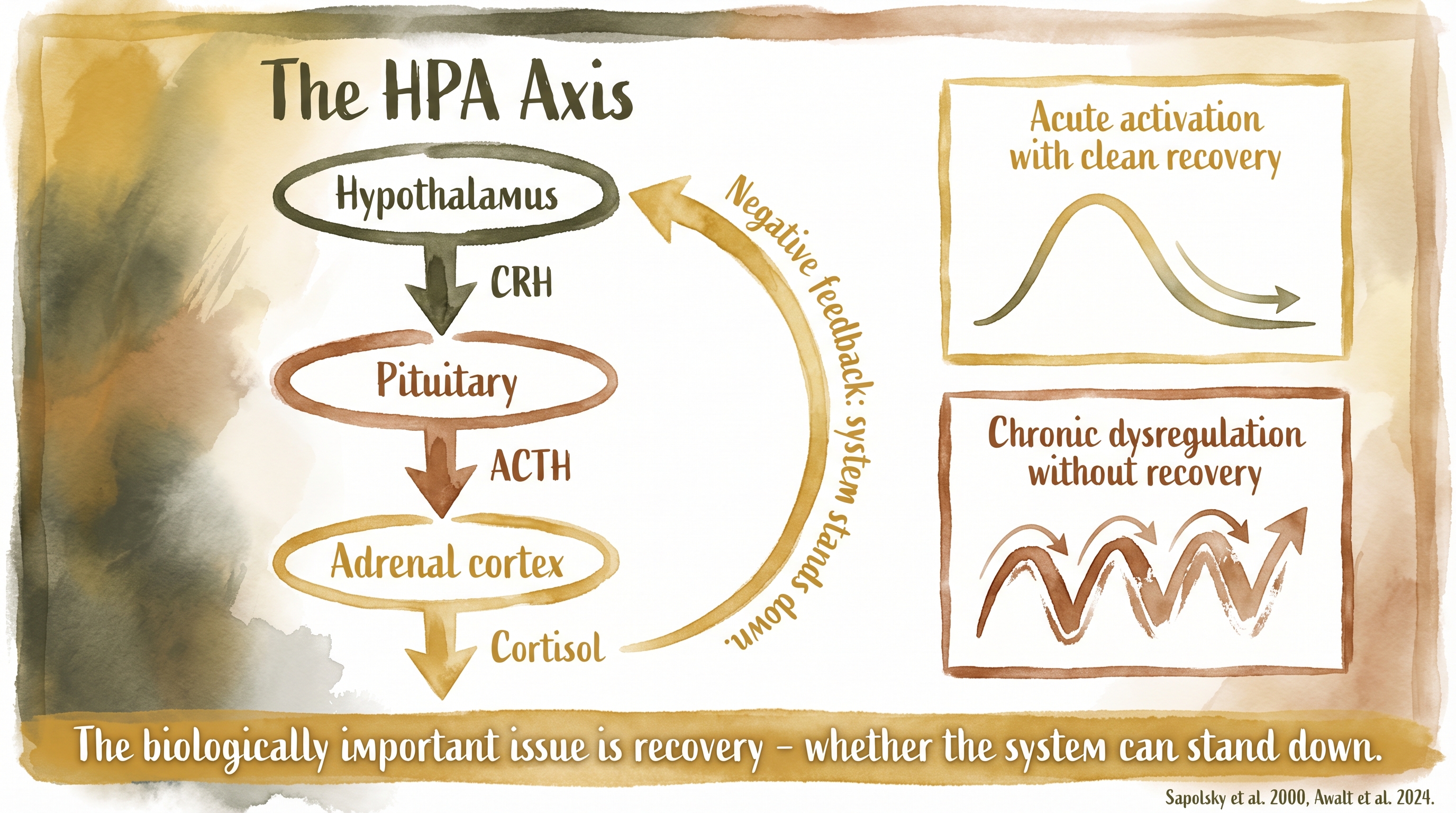
The HPA axis translates perceived threats into cortisol release through a cascade from hypothalamus to adrenal cortex.
Key Takeaways
- The canine HPA axis is a conserved endocrine stress system built around CRH, ACTH, and cortisol.
- Acute activation is adaptive; the biologically important issue is whether the system recovers cleanly.
- Early experience can alter the later shape of canine stress regulation, with direct dog evidence now available for epigenetic association.
- Chronic dysregulation is linked to downstream immune and welfare-relevant consequences, especially in adverse or unstable environments.
The Evidence
- Stress-immunity synthesisdomestic dogs
Dogs use cortisol as the primary glucocorticoid in the HPA response, and puppies as young as seven weeks show measurable salivary cortisol increases under acute challenge. - Awalt, S. L. et al. (2024)domestic dogs
Early-life adversity in dogs was associated with altered NR3C1 and OXTR methylation, cortisol reactivity differences, and attachment-related variation. - Beerda, B. et al. (1999)domestic dogs
Chronic social and spatial restriction altered behavioral, hormonal, and immunological responses in dogs. - Hennessy, M. B. et al. (1997) and van der Laan, J. E. et al. (2022)domestic dogs
Shelter intake and rehoming contexts produced marked cortisol disruption, with improvement over time in quieter and more stable environments.
- Weaver, I. C. G. et al. (2004)rats
Maternal care altered glucocorticoid-related methylation patterns and later stress reactivity through environmental rather than purely genetic pathways. - Sapolsky, R. M. et al. (2000)multiple mammals
Established the central role of glucocorticoids and feedback regulation in mammalian stress physiology.
- SCR-094 boundarydomestic dogs
Early-life-associated methylation changes in dogs are documented, but the full causal chain from those marks to specific lifelong behavioral or health outcomes should be stated more cautiously. - SCR-045 boundarydomestic dogs
Immune consequences are documented under chronic adverse-environment stressors, but should not be casually generalized to every household arousal pattern without qualifier.
No published study has directly measured early-life HPA-axis calibration in puppies from the same litter living in different caregiving environments, making it impossible to quantify the effect size of early environment on stress physiology.
No study has tracked longitudinal HPA-axis function in dogs from puppyhood through old age to measure stability of early-life differences or capacity for later reorganization.
SCR References
Sources
- Awalt, S. L., et al. (2024). A dog's life: Early life histories influence methylation of glucocorticoid (NR3C1) and oxytocin (OXTR) receptor genes, cortisol levels, and attachment styles. Developmental Psychobiology.
- Beerda, B., et al. (1999). Chronic stress in dogs subjected to social and spatial restriction. II. Hormonal and immunological responses. Physiology & Behavior.
- Hennessy, M. B., et al. (1997). Plasma cortisol levels of dogs at a county animal shelter. Physiology & Behavior.
- Sapolsky, R. M., Romero, L. M., & Munck, A. U. (2000). How do glucocorticoids influence stress responses? Endocrine Reviews, 21(1), 55-89.
- van der Laan, J. E., et al. (2022). Evaluation of hair cortisol as an indicator of long-term stress responses in dogs in an animal shelter and after subsequent adoption. Scientific Reports, 12, 5117.
- Weaver, I. C. G., Cervoni, N., Champagne, F. A., D'Alessio, A. C., Sharma, S., Seckl, J. R., Dymov, S., Szyf, M., and Meaney, M. J. (2004). Epigenetic programming by maternal behavior. Nature Neuroscience, 7(8), 847-854. DOI: 10.1038/nn1276.