Epigenetic Programming in Early Life
Compound evidence detail2 SCRs / 4 parts
- Documentedthe canine methylation pattern reported by Awalt 2024 for NR3C1 and OXTR in dogs with measured early-life history
- Heuristicthe JB extrapolation that ordinary calm raising produces durable epigenetic advantages in dogs beyond preventing adverse environments
- Documentedthe Awalt 2024 finding that NR3C1 and OXTR methylation in dogs covaries with documented early-life experience
- Heuristicthe JB inference that those documented methylation differences in dogs predict specific later health or developmental outcomes
Early environment can change more than behavior. It can also alter how stress- and bonding-related genes are regulated. That claim is documented in rats through classic maternal-care epigenetics and now has direct canine support through work on NR3C1 and OXTR methylation. The bridge is real, but the bridge still has species boundaries. Documented-Cross-Species
What It Means
Weaver and Meaney's rat work made the basic mechanism famous. Rat mothers who showed high licking and grooming produced offspring with altered glucocorticoid-receptor expression through methylation changes at NR3C1. Documented-Cross-Species Cross-fostering showed the effect was environmental rather than purely genetic. That is why the paper still matters so much. It demonstrated a path by which caregiving style could become molecularly legible inside the offspring stress system.
The dog literature used to stop at plausibility. Documented-Cross-Species It no longer does. Awalt et al. (2024) found that dogs with more adverse early-life histories differed in methylation on NR3C1 and OXTR, with linked cortisol and attachment-related differences. That does not replicate the rat protocol one for one. The independent variable in the dog study was broad early-life adversity rather than a tightly controlled maternal-tactile behavior. But it does close the biggest old objection, which was that the epigenetic programming story had never been shown directly in dogs at all.
That leaves the correct ceiling in a more interesting place. The general principle that early environment can alter epigenetic marks on stress and bonding genes is now documented in dogs. Documented The more specific JB claim, that breeder-period calmness and early family continuity write a particular protective molecular code in a reliably predictable way, is still more interpretive. The species gap is smaller than it used to be, but the intervention map is still incomplete.
One helpful way to hold the science is this: the environment does not have to change DNA sequence to change developmental trajectory. It can change what parts of the stress and bonding system are more or less readily expressed. Documented That is what makes epigenetic programming so relevant to the Foundations layer. It shows that early environment is not only remembered by the dog. It can become part of the biological settings through which later life is processed.
Why It Matters for Your Dog
For families, this entry raises the stakes on early environment without becoming melodramatic about it. Calmness, predictability, and chronic adversity are not simply momentary experiences. They can contribute to the regulatory backdrop through which the puppy's stress and bonding systems develop. Documented
The epigenetic literature helps explain why JB treats calm early environment as a developmental condition rather than as a cosmetic preference. Early life can shape how the stress system is regulated from the inside.
This does not mean a family can engineer a puppy's molecular future with perfect precision. It does mean the older idea that early handling and atmosphere only matter behaviorally is no longer strong enough. Dogs now have direct evidence that early-life history reaches the level of gene regulation.
That is one reason the breeder period matters so much in JB. The dog is not arriving as a blank slate at eight or twelve weeks. By then, the stress and bonding systems have already been living inside an environment long enough for early programming effects to be scientifically plausible and, in some cases, directly measurable.
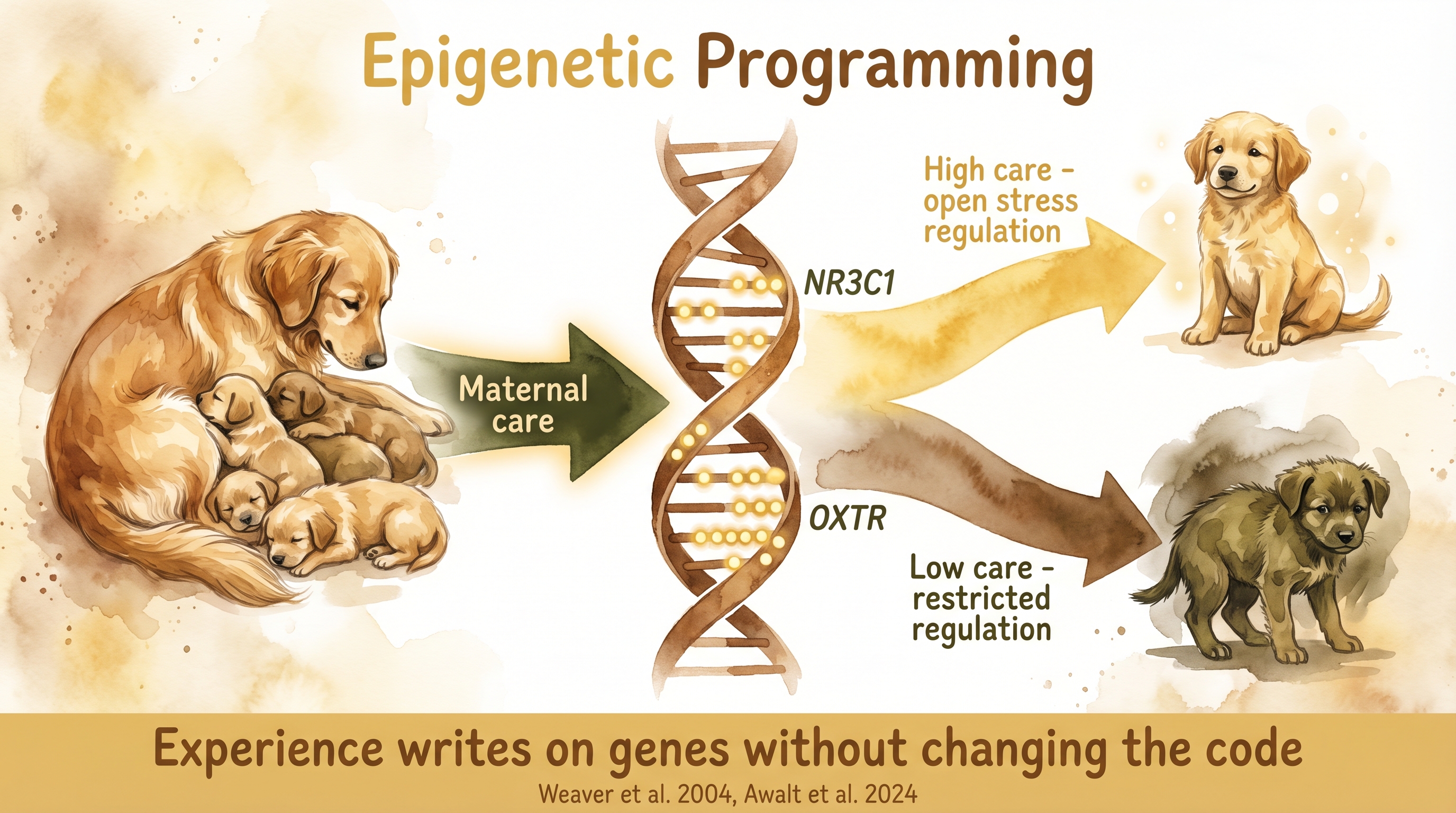
Early caregiving writes on genes without changing the code - maternal care alters methylation patterns that shape stress regulation and attachment.
Key Takeaways
- Epigenetic programming means early environment can change gene regulation without changing DNA sequence.
- The classic rat evidence showed that caregiving style can alter stress-gene expression through methylation.
- Dogs now have direct evidence too: early-life adversity is associated with altered NR3C1 and OXTR methylation.
- The broad principle is documented, but the exact map from JB-style practices to specific canine methylation outcomes is still incomplete.
The Evidence
- Weaver, I. C. G. et al. (2004)rats
Showed that maternal licking and grooming altered glucocorticoid-related methylation and receptor expression, with cross-fostering confirming the effect was environmental rather than purely genetic. - Meaney, M. J. and related rat workrats
Built the mechanistic framework linking early caregiving, methylation, and later stress regulation.
- Awalt, S. L. et al. (2024)domestic dogs
Found associations between early-life history and altered methylation on NR3C1 and OXTR, with linked cortisol and attachment-related differences. - SCR-094 synthesisdomestic dogs
Confirms that early-life stress alters epigenetic marks on stress and bonding genes in dogs, even though many downstream health and behavior consequences remain less fully mapped.
No published canine study has yet isolated which exact early caregiving variables, such as calm handling, predictable routine, or breeder-family continuity, produce which methylation outcomes. The current evidence documents the principle more strongly than the intervention details.
SCR References
Sources
Weaver, I. C. G., Cervoni, N., Champagne, F. A., D'Alessio, A. C., Sharma, S., Seckl, J. R., Dymov, S., Szyf, M., & Meaney, M. J. (2004). Epigenetic programming by maternal behavior. Nature Neuroscience, 7(8), 847-854. https://doi.org/10.1038/nn1276
Awalt, S. L., Boghean, L., Klinkebiel, D., & Strasser, R. (2024). A dog's life: Early life histories influence methylation of glucocorticoid (NR3C1) and oxytocin (OXTR) receptor genes, cortisol levels, and attachment styles. Developmental Psychobiology, 66(8), e22482. https://doi.org/10.1002/dev.22482
Meaney, M. J. (2001). Maternal care, gene expression, and the transmission of individual differences in stress reactivity across generations. Annual Review of Neuroscience, 24, 1161-1192. https://doi.org/10.1146/annurev.neuro.24.1.1161
Szyf, M., Weaver, I., & Meaney, M. (2007). Maternal care, the epigenome and phenotypic differences in behavior. Reproductive Toxicology, 24(1), 9-19. https://doi.org/10.1016/j.reprotox.2007.05.001