Canine Epigenetic Evidence
Compound evidence detail2 SCRs / 4 parts
- Documentedthe Awalt 2024 finding that NR3C1 and OXTR methylation in dogs covaries with documented early-life experience
- Heuristicthe JB inference that those documented methylation differences in dogs predict specific later health or developmental outcomes
- Documentedthe canine methylation pattern reported by Awalt 2024 for NR3C1 and OXTR in dogs with measured early-life history
- Heuristicthe JB extrapolation that ordinary calm raising produces durable epigenetic advantages in dogs beyond preventing adverse environments
The dog-specific epigenetics literature is still small, but it is no longer empty. The strongest direct evidence shows that early-life adversity in dogs is associated with altered methylation on genes relevant to stress regulation and bonding, especially NR3C1 and OXTR. What the literature does not yet provide is a large, intervention-based map showing which precise caregiving practices generate which methylation outcomes. Documented
What It Means
SCR-094 is the central entry for this page. Awalt and colleagues found that dogs with adverse early-life histories differed in methylation on NR3C1 and OXTR, and that these patterns were associated with cortisol and attachment-related measures. That is a meaningful canine finding because it directly links developmental history with molecular markers in a dog sample.
The literature around canine aging also supports a broader point: the canine epigenome is measurable, informative, and responsive enough to be used in aging-clock and biological-timing research. Documented That does not make every developmental claim proven, but it reinforces that epigenetic analysis in dogs is scientifically real rather than speculative.
Why the Dog Literature Still Feels Thin
Compared with the classic rodent literature, dog epigenetic work is still limited by small samples, relatively few longitudinal designs, broad environmental categories rather than tightly controlled caregiving variables, and incomplete tissue and outcome mapping. Documented This is not a reason to dismiss the findings. It is a reason to keep them precise.
What the Evidence Supports About Dogs
The direct dog evidence supports these claims most comfortably: the canine epigenome is responsive to developmental history, stress- and bonding-related genes can show methylation differences associated with early-life adversity, and these differences can co-occur with cortisol and attachment-related measures. Documented
The evidence is much less comfortable supporting: one exact family practice causes one exact methylation outcome, calm household structure has already been proven to produce a specific protective canine epigenetic pattern, or methylation findings by themselves fully explain later adult temperament. Documented
Breed, Age, and Developmental Relevance
One reason the field will likely grow is that dogs are unusually useful for developmental and comparative work. Breed histories, structured pedigrees, aging-clock research, and environmentally varied life conditions make them scientifically interesting models. Documented
At the same time, those same differences complicate interpretation. Breed effects, age effects, tissue effects, and living-environment effects can all influence what methylation findings mean. That is why responsible dog epigenetics writing should sound more like "evidence of developmental sensitivity exists" and "mechanistic follow-up is still building" rather than "the epigenetic dog story is already complete."
Why It Matters for Your Dog
The main value of the canine literature is not that it proves every larger theory. It is that it removes the idea that dog developmental epigenetics is purely hypothetical. Dogs now have direct evidence in this space. That means families can responsibly understand that the early environment is molecularly relevant, while still being careful about exactly how far the current literature goes.
The maternal-care layer uses this science to argue that early environment leaves more than a behavioral memory. The dog literature now supports that general direction, while still leaving most practice-specific intervention claims more conservative.
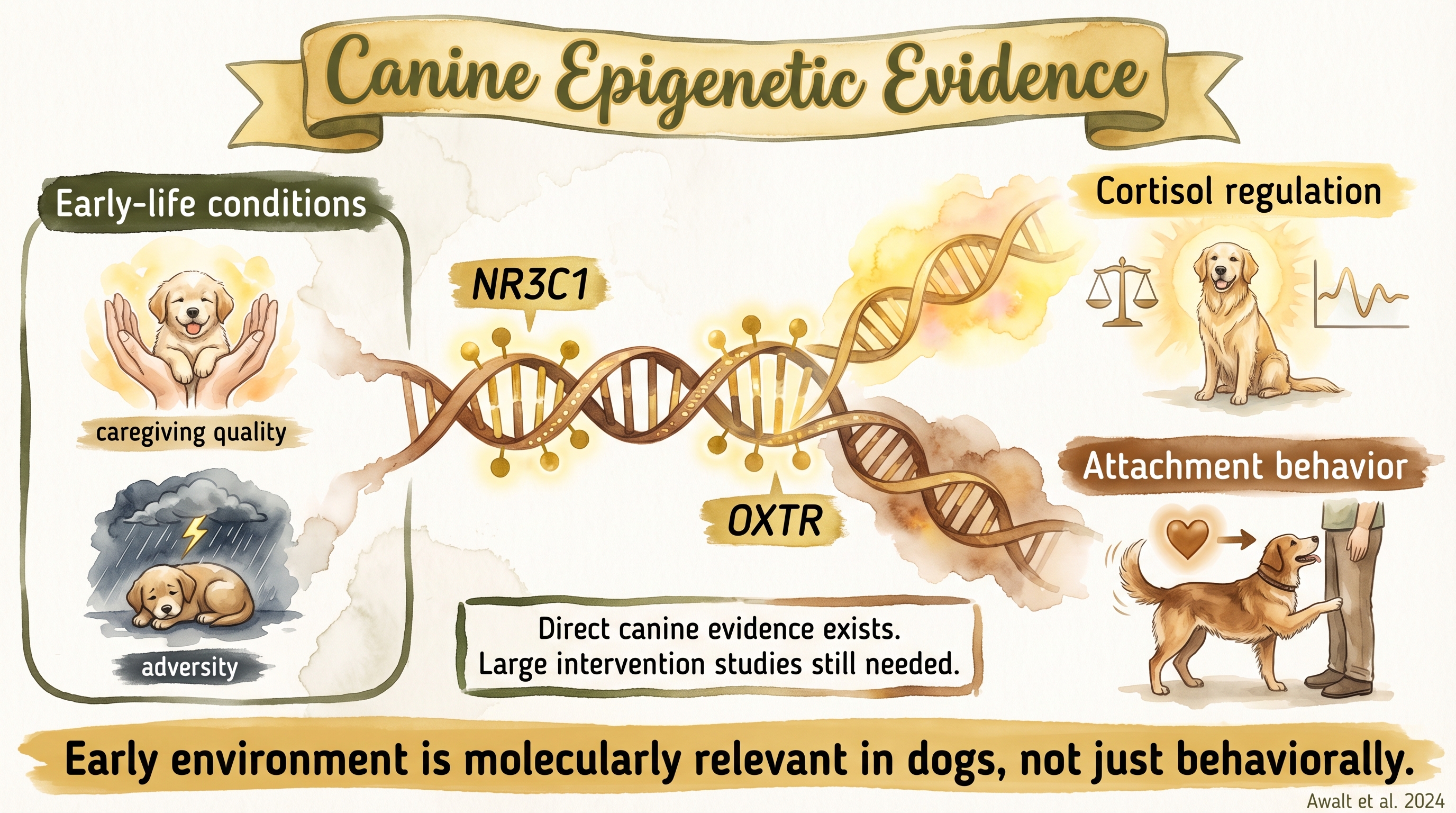
Early environment shapes gene expression in dogs through methylation changes at stress-response loci.
Key Takeaways
- Dog-specific epigenetic evidence is limited but real.
- The strongest canine findings currently involve NR3C1 and OXTR methylation associated with early-life adversity.
- The literature supports developmental sensitivity more strongly than practice-specific intervention claims.
- Canine epigenetics now belongs in serious developmental discussion, but it still requires careful ceilings.
The Evidence
- Awalt, S. L. et al. (2024)domestic dogs
Reported altered NR3C1 and OXTR methylation associated with early-life adversity, cortisol response, and attachment-related measures. - Canine epigenetic clock and aging literaturedomestic dogs
Supports the canine epigenome as measurable and biologically meaningful, even outside stress-focused developmental studies.
- Foundational rodent and human stress-epigenetics literaturerats and humans
Provides the larger mechanistic context that makes the canine findings scientifically interpretable rather than isolated curiosities.
- SCR-094 boundarydomestic dogs
The evidence supports epigenetic responsiveness of the canine stress system to early-life history, but not a detailed intervention map showing which exact JB-style practices produce which methylation outcomes.
No published study has measured whether specific caregiving practices such as structured calm raising produce measurable changes in canine NR3C1 or OXTR methylation, leaving the intervention-to-methylation bridge unmapped.
No longitudinal study in dogs has tracked whether early-life epigenetic marks on stress genes predict behavioral or health outcomes in adulthood, limiting current findings to association rather than prediction.
SCR References
Sources
- Awalt, S. L., et al. (2024). A dog's life: Early life histories influence methylation of glucocorticoid (NR3C1) and oxytocin (OXTR) receptor genes, cortisol levels, and attachment styles. Developmental Psychobiology.
- Canine epigenetic aging source package: retain as bounded synthesis unless a specific clock or aging primary source is chosen for this entry.
- Foundational stress-epigenetics source package: rodent and human mechanisms support biological plausibility but do not verify normal-range JB caregiving effects in dogs.
- SCR-094 boundary: Awalt 2024 verifies early-life adversity associations with dog blood methylation; normal-range caregiving and brain-tissue claims remain outside documented evidence.