Epigenetics: An Overview
Compound evidence detail2 SCRs / 4 parts
- Documentedthe canine methylation pattern reported by Awalt 2024 for NR3C1 and OXTR in dogs with measured early-life history
- Heuristicthe JB extrapolation that ordinary calm raising produces durable epigenetic advantages in dogs beyond preventing adverse environments
- Documentedthe Awalt 2024 finding that NR3C1 and OXTR methylation in dogs covaries with documented early-life experience
- Heuristicthe JB inference that those documented methylation differences in dogs predict specific later health or developmental outcomes
Epigenetics is the study of relatively stable changes in gene expression that occur without changing the DNA sequence itself. The underlying genome stays the same, but the way genes are read, amplified, or quieted can change over development and in response to environment. In dogs, the strongest current evidence shows that early-life adversity is associated with altered methylation on stress- and bonding-related genes. The broader mechanistic foundation is much older and much deeper in rodent and human work. Documented
What It Means
Genes are not destiny in a simple one-gene, one-outcome sense. Cells need ways to decide which parts of the genome should be more active, less active, or temporarily silenced under different developmental and environmental conditions. That regulatory layer is what epigenetics is about.
The major mechanisms include DNA methylation, histone modification, and non-coding RNA regulation. This dispatch focuses mainly on DNA methylation because it is the best-established mechanism in the stress-regulation literature relevant to dogs. Documented
Stress biology is not only about the immediate cortisol response. It is also about how stress systems get calibrated over time. Epigenetics matters here because it offers a plausible molecular bridge between early caregiving, adversity or predictability, later stress reactivity, and longer-term attachment and regulation differences. This is one reason the field matters so much to developmental theory. It gives a serious biological vocabulary for how environment can have durable effects without changing inheritance at the DNA-sequence level.
The Foundational Rodent Work
The classic starting point is Meaney and colleagues' maternal-care research in rats. High licking and grooming were associated with lower methylation at glucocorticoid-receptor regulatory regions, higher receptor expression, and more efficient HPA-axis negative feedback in offspring. Cross-fostering showed that the effect was environmental rather than simply genetic. Documented That work became foundational because it showed, in a controlled model, that early caregiving could become a molecular developmental variable. This remains one of the most important places where wiki writing must stay careful. The rat evidence is strong. The canine extension is not identical in depth, method, or certainty.
The Human Extension
Human work extended the same general logic. McGowan and colleagues found altered NR3C1 methylation in hippocampal tissue from suicide completers with histories of childhood abuse compared with controls. That did not prove that every later-life outcome can be reduced to one epigenetic mechanism, but it strengthened the idea that early adversity and stress-regulation genes can remain linked in the human nervous system as well. Documented Together, the rodent and human literature make the mechanistic framework highly credible across mammals.
Why It Matters for Your Dog
Dogs now have direct evidence in this area, and SCR-094 is the key entry. Awalt and colleagues reported that early-life adversity in dogs was associated with altered methylation on NR3C1 (the glucocorticoid receptor gene) and OXTR (the oxytocin receptor gene). Those molecular differences were associated with cortisol reactivity and attachment-related outcomes. Documented That is important because it moves canine epigenetics from pure extrapolation to direct evidence.
The dog finding is still narrower than the older rodent story in several important ways: the sample sizes are smaller, the environmental categories are broader, the designs are less experimentally controlled, and the downstream causal chain is less fully mapped. So the safe summary is not "dogs have the whole Meaney mechanism proven." The safe summary is "dogs now have direct evidence that early life is associated with methylation differences in stress- and bonding-related genes."
What the Field Can and Cannot Yet Say
Epigenetics often sounds more deterministic than it should. The field can responsibly say that early environment can be associated with durable shifts in gene regulation, these shifts can involve genes relevant to stress and bonding, and dogs show direct evidence of this association. Documented
The field should be more careful about saying: one specific raising style permanently writes one exact lifelong outcome, methylation differences alone explain complex temperament or health outcomes, or canine caregiving interventions have already been shown to produce a known protective epigenetic pattern at scale.
That last claim is especially relevant to JB. The project-level interpretation that calm, structured raising produces protective epigenetic architecture is biologically plausible and directionally consistent with the literature. It is not yet a directly tested canine intervention result.
Why the Canine Evidence Still Matters
Even with those boundaries, the current dog evidence is a real milestone. It supports a scientifically serious statement that early-life history in dogs is not only behaviorally relevant. It is molecularly relevant. That changes how developmental claims should be framed. The early environment is not simply shaping habits from the outside. It may also be participating in how stress-regulation systems are set and expressed over time.
Epigenetics Is Not a One-Way Fate
Another reason caution matters is that epigenetics is often heard as a story of permanent damage or permanent advantage. The real picture is more nuanced. Some marks become relatively stable. Some remain plastic. Some depend on age, tissue, or context. Dog evidence itself suggests age-sensitive methylation variation rather than a perfectly frozen developmental script.
This page is strongest when it frames epigenetics as a mechanism of developmental influence, a source of durable but not necessarily immutable difference, and a field that supports probabilistic calibration more than fatalistic destiny.
The calmness layer often argues that early environment gets into the dog at a deep biological level. Epigenetics helps explain why that is a serious scientific claim, while also requiring discipline about how directly current canine evidence maps onto specific JB-style raising practices.
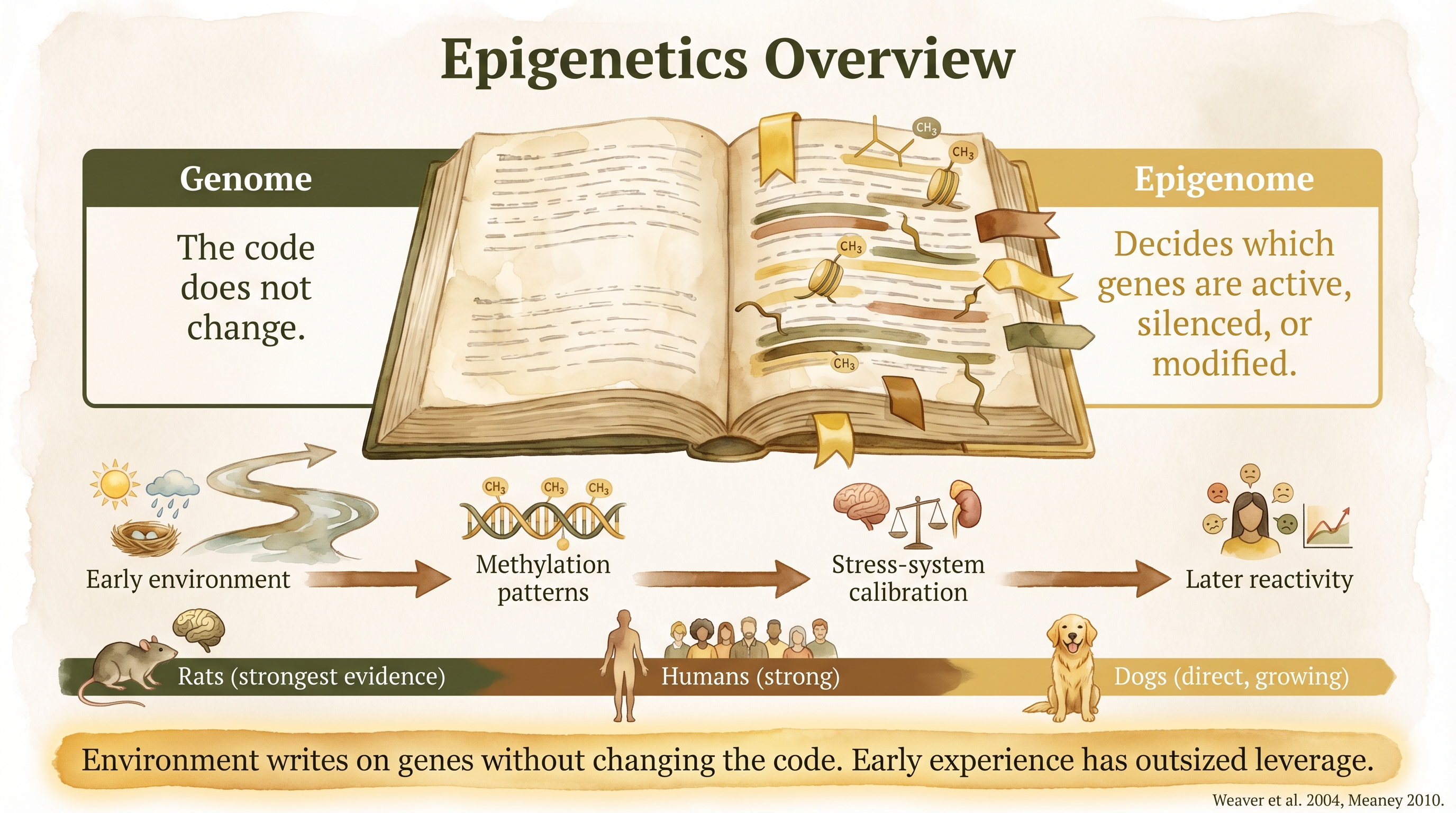
Epigenetics explains how environment regulates gene expression through methylation, histone modification, and RNA pathways.
Key Takeaways
- Epigenetics describes durable changes in gene expression without changing DNA sequence.
- The strongest mechanistic foundation comes from rodent and human stress-regulation research.
- Dogs now have direct evidence linking early-life history with methylation differences in stress- and bonding-related genes.
- The field supports developmental calibration, but not a simplistic destiny story or a fully proven JB-specific intervention claim.
The Evidence
- Weaver, I. C. G. et al. (2004)rats
Showed that maternal care altered methylation at glucocorticoid-receptor regulatory regions and changed later stress physiology in offspring. - Meaney, M. J. (2005, 2010)rats and mammalian developmental synthesis
Framed epigenetic programming as a bridge between early environment and later stress regulation. - McGowan, P. O. et al. (2009)humans
Found altered NR3C1 methylation in hippocampal tissue associated with histories of childhood abuse.
- Awalt, S. L. et al. (2024)domestic dogs
Reported early-life-history associations with altered NR3C1 and OXTR methylation, alongside cortisol and attachment-related differences in dogs. - Stress-immunity source synthesisdomestic dogs
Supports the canine epigenome as responsive to developmental history while leaving causal downstream consequence claims more cautious.
- SCR-094 boundarydomestic dogs
The documented dog finding is early-life-associated methylation change. The stronger claim that calm raising produces a known protective methylation profile in puppies remains an interpretive bridge rather than a direct intervention result.
No published study has measured canine epigenetic marks before and after a documented caregiving intervention, making it impossible to claim that any specific raising practice produces a known methylation outcome.
No study has tracked the same dogs' epigenetic marks from early puppyhood through adulthood to measure stability, reversibility, or long-term functional consequence.
SCR References
Sources
- Awalt, S. L., et al. (2024). A dog's life: Early life histories influence methylation of glucocorticoid (NR3C1) and oxytocin (OXTR) receptor genes, cortisol levels, and attachment styles. Developmental Psychobiology.
- McGowan, P. O., et al. (2009). Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse. Nature Neuroscience, 12(3), 342-348.
- Meaney, M. J. (2005). Environmental programming of stress responses through DNA methylation. Dialogues in Clinical Neuroscience.
- Meaney, M. J. (2010). Epigenetics and the biological definition of gene x environment interactions. Child Development, 81(1), 41-79.