Hereditary Cancer Risk in Golden Retrievers
Cancer is one of the hardest truths in Golden Retriever breeding because it is both a breed-level burden and a scientifically difficult target, and the combination makes it almost uniquely challenging to talk about honestly. The literature supports that Goldens carry an unusually heavy cancer load relative to many breeds and that inherited risk is part of that story. The same literature also makes clear that this is not a single-gene problem with a clean DNA fix. It is a polygenic, multifactorial burden that thoughtful breeding can influence but not currently solve, and pretending otherwise in either direction (minimizing the burden or promising a fix that does not exist) fails families who deserve straight talk about what the science actually shows. Documented
What It Means
The breed-level picture
Golden Retrievers appear in multiple datasets as a cancer-heavy breed, and the consistency of that finding across different study methodologies is part of why the conclusion is robust even though the exact numbers vary. Documented Depending on the dataset and the denominator used, cancer may account for a very large share of deaths in the breed, often at a level that makes it impossible to dismiss as just ordinary large-breed aging. Studies looking at cause of death in Golden Retriever populations have produced estimates suggesting that cancer is responsible for more than half of deaths in the breed in some analyses, which places Goldens at or near the top of breed-specific cancer burden rankings in the broader canine epidemiology literature.
At the same time, cancer percentages vary sharply depending on whether the study population is a referral necropsy series (which overrepresents cases that ended up at specialist facilities), an insurance dataset (which reflects claims from the subset of dogs whose caregivers have insurance and who sought treatment), a caregiver survey (which depends on caregiver recall and categorization accuracy), or a prospective cohort (which follows dogs forward in time and typically produces the most rigorous data). Observed-JB Each of these designs has different biases, and the exact percentage any one study reports should be understood in the context of its methodology rather than treated as the single true number.
That methodological variation matters because it changes the exact figures that get quoted in breeder conversations and family-facing materials. It does not erase the broader conclusion, which holds across study types even though the specific numbers differ. Golden Retrievers are burdened by cancer at a level breeders have to treat as central to any honest program, not incidental, and any breeder who downplays the burden to protect marketing language is not serving the breed or the families considering it.
The specific cancers that matter
The main breed-relevant cancers usually discussed in Golden Retriever contexts fall into a few dominant categories. Hemangiosarcoma is a cancer of the cells that line blood vessels and can appear in various locations, with splenic and cardiac forms being particularly devastating because they often present with sudden collapse from internal hemorrhage rather than with warning signs that would allow earlier intervention. Lymphoma is a cancer of the lymphatic system that is also common in the breed and tends to have a somewhat more treatable profile in the short term but rarely achieves durable cure. Mast cell tumors affect a type of immune cell and range from easily removed low-grade lesions to aggressive high-grade cancers that can metastasize. Osteosarcoma is a bone cancer more strongly associated with large and giant breeds generally, and Golden Retrievers sit in a size range where it contributes to the overall cancer picture without being the dominant concern that hemangiosarcoma and lymphoma represent.
These cancers are not all the same disease at the biological level, and their different molecular mechanisms mean that any genetic analysis has to respect the differences between them rather than treating "cancer" as one uniform category. A dog from a line with heavy hemangiosarcoma history may or may not be at elevated risk for lymphoma, and the two cancers have distinct enough genetic signatures that the risk profiles are partly independent. Documented
Why this becomes a breeding-genetics issue
The reason cancer burden becomes a breeding-genetics issue, rather than just an epidemiological observation, is that these cancers cluster at the breed and line level strongly enough to support inherited liability as part of the explanation. If cancer were purely an environmental or stochastic phenomenon, we would expect it to be distributed roughly uniformly across lines within a breed, and the observed reality is that certain lines have heavier cancer histories than others. That line-level clustering is suggestive of inherited contribution, and the broader epidemiological comparison between breeds (with some breeds showing much lower cancer burden than Goldens in comparable environments) is also consistent with the view that breed genetics contributes meaningfully to the Golden cancer burden.
But the inherited liability is not Mendelian in the way PRA is Mendelian. The risk is polygenic and probabilistic, meaning that many genetic factors each contribute small amounts of liability and that the total genetic contribution is distributed across the genome rather than concentrated at a few big-effect loci. This polygenic architecture is why attempts to develop simple predictive DNA tests for Golden Retriever cancer risk have not produced the clean clear-carrier-affected sorts that families sometimes ask for. The biology does not currently support that kind of test, and the absence of such a test is not a failure of breeder effort but a reflection of how complex traits with multifactorial etiology actually behave.
What breeders can actually do
That means there is no current tool that lets a breeder say with confidence "this dog will not get cancer" or even "this line has zero cancer risk." What the breeder can do is track line history carefully across multiple generations so that the accumulated cancer experience of the pedigree is known rather than hidden, preserve diversity so that polygenic risk factors are not concentrated through inbreeding on narrow portions of the gene pool, avoid repeating especially burdened combinations when line history suggests certain crosses have produced unusual cancer clustering, and use published risk literature honestly rather than cherry-picking the most optimistic interpretation for marketing purposes.
Each of these practices is less satisfying than a simple screening test would be, and none of them produces the kind of certainty families sometimes hope for when they ask about cancer risk. But each is a real contribution to managing probabilities at the line level, and the cumulative effect of careful stewardship over generations is not trivial even though it cannot be expressed in a single test result.
The Golden Retriever Lifetime Study
The Golden Retriever Lifetime Study matters in this context because it is the most ambitious prospective attempt to map how genetics, environment, and life history interact in the breed's cancer burden. The study has followed thousands of Golden Retrievers from early life forward, collecting extensive data on diet, environment, medical history, and genetic samples, with the goal of identifying risk factors that can inform both breeding and lifestyle decisions. The data generated by this project is one of the most comprehensive canine longitudinal datasets ever assembled, and the insights that emerge from it over the coming years will likely shape the breed's cancer conversation in important ways.
But even that project does not magically convert the problem into one clean screening answer. Longitudinal studies produce statistical associations and risk factors, not deterministic predictions, and the translation from research findings to individual breeding decisions is always going to involve probabilistic reasoning rather than simple sorting. Documented Breeders and families who follow the Lifetime Study's output will find it valuable for refining their understanding of risk, but they should not expect it to produce a single decisive test that eliminates the need for careful line-level stewardship.
The honesty problem
Cancer in Goldens is also a case where communication discipline matters as much as the underlying science. It is tempting for breeders to either downplay the burden (because the numbers are frightening and scare away prospective families) or dramatize it (because invoking cancer can signal seriousness about health management). Both failures produce distorted conversations. Minimizing the burden misleads families and caregivers who are making major decisions about adding a dog to their lives. Dramatizing it pushes them toward fatalism that can undermine the real benefit of careful breeding and diversity management.
The honest middle position is harder to maintain but more useful. Cancer burden in Goldens is real and substantial. Inherited contribution is part of the picture. Current tools cannot solve the problem in one generation. Careful stewardship still matters because probability is not destiny and the alternative (neglect) would make the breed's situation worse rather than better.
Why It Matters for Your Dog
Families deserve honesty here because this is one of the most emotionally consequential breed facts, and the quality of the conversation a family has with their breeder about cancer risk is often one of the clearest signals of whether the breeder is approaching the program with the integrity the breed requires.
The wrong communication style either minimizes the burden in ways that leave families underprepared for the reality of breed ownership, or dramatizes it so heavily that no rational breeder choice seems meaningful and families walk away either paralyzed or cynical. Both responses are failures of the breeder's responsibility to inform, and both can be recognized by the language breeders use when the topic comes up. A breeder who waves off cancer concerns with "oh, all breeds get cancer" is minimizing. A breeder who shrugs and says "there is nothing we can do, it is just the breed" is dramatizing into fatalism. Neither is telling the family the truth the literature actually supports.
The right communication style is specific and balanced. Yes, cancer burden in the breed is real and substantial, and families should expect to face this as part of Golden Retriever ownership. Yes, inheritance matters, which is why line history and diversity stewardship are part of responsible breeding even without a definitive test. No, current testing does not solve the problem in one generation, and breeders who claim otherwise are overclaiming. Yes, careful breeding, diversity stewardship, and attention to line history still matter because they shift the probability distribution favorably even if they do not eliminate the risk.
Families who hear this honest version are better equipped to partner with the breeder over the dog's life, to take cancer screening seriously as the dog ages, and to hold reasonable expectations rather than swinging between false reassurance and fatalism. That is the kind of informed partnership the breed needs, and it starts with breeders willing to have the difficult conversation rather than deflecting it.
For JB, this means cancer cannot be treated as background noise or as a topic to be handled only when families ask. It has to be part of the breeding conversation even when the science is frustratingly incomplete compared with simple Mendelian disease management, and the program's willingness to discuss cancer honestly is part of what distinguishes a serious approach from a cosmetic one. Observed-JB
What This Cannot Predict
Cancer heritability in Goldens cannot tell you which specific dog will get cancer, because the polygenic and multifactorial architecture leaves substantial individual variation around any line-level estimate. Documented
It cannot tell you when cancer will appear, because age at onset varies across cancer types and individual dogs even within lines of similar overall risk.
It cannot make a line with favorable history immune, because polygenic risk is never fully captured by the visible pedigree and because environmental and stochastic factors continue to contribute even in the best-managed lines.
And it cannot justify casual catastrophe language about every Golden puppy as though the breed were beyond help. The burden is real, but so is the variation, and fatalism is not warranted by the current evidence any more than minimization is.
The correct statement is narrower and stronger than either of those extremes: Golden Retrievers carry a documented breed-level cancer burden with a real inherited component, and current tools are better at managing probabilities at the line level than at guaranteeing outcomes for any individual puppy.
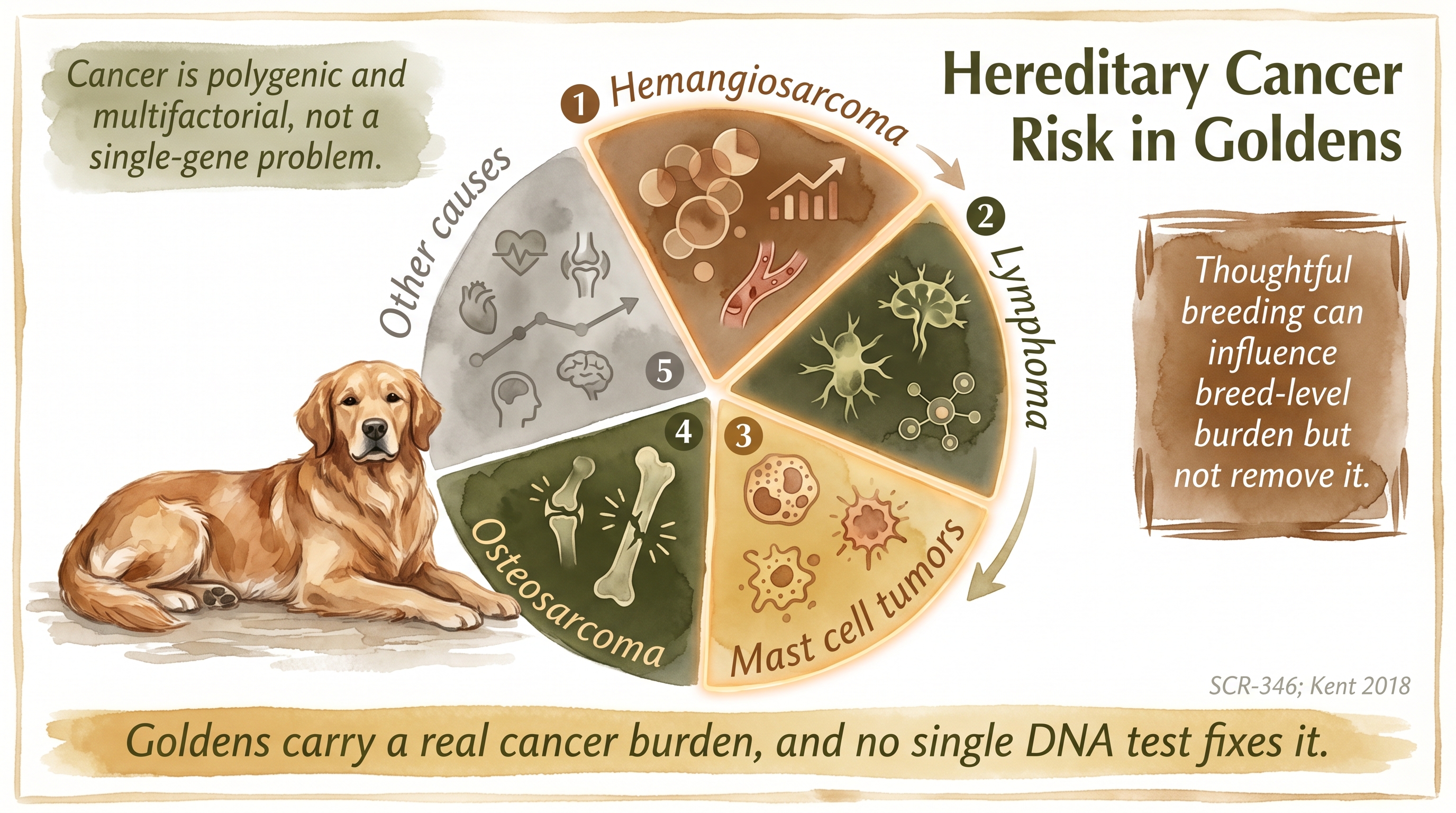
Goldens carry a real cancer burden, and no single DNA test fixes it.
Key Takeaways
- Golden Retrievers carry a documented breed-level cancer burden that breeders cannot responsibly ignore.
- The inherited component is real, but it is polygenic and multifactorial rather than a simple Mendelian problem.
- Current tools help manage probabilities and line-level choices, not guarantee that a given dog will remain cancer-free.
- Honest breeder communication should neither minimize the burden nor turn it into fatalistic catastrophe language.
- Line history, diversity stewardship, and ongoing research participation remain the main practical levers for managing risk.
The Evidence
This entry uses observed claim-level tags beyond the dedicated EvidenceBlocks below. These tags mark JB program observation or practice-derived claims that need dedicated EvidenceBlock coverage in a later content pass.
- Golden Retriever cancer epidemiology literatureGolden Retrievers
Golden Retrievers show a heavy cancer burden across multiple datasets, with cancer representing a dominant cause of death in important breed-specific study populations. - Golden Retriever Lifetime Study and related epidemiologyGolden Retrievers
Prospective and retrospective work supports that cancer is a central breed-health problem and that both inherited and environmental contributors are under active investigation.
- Golden Retriever oncology genetics literatureGolden Retrievers
Cancer susceptibility in the breed shows inherited structure, but current evidence supports a polygenic and multifactorial picture rather than a single-gene predictive model.
No published genome-wide association study has yet validated a polygenic risk score for Golden Retriever cancer that shows high individual-level predictive validity in an independent prospective cohort.
SCR References
Sources
- Kent M.S., Burton J.H., Dank G., Bannasch D.L., & Rebhun R.B. (2018). Association of cancer-related mortality, age and gonadectomy in golden retriever dogs at a veterinary academic center (1989-2016). PLOS ONE, 13(2), e0192578. doi:10.1371/journal.pone.0192578
- Bonnett B.N., Egenvall A., Hedhammar Ã…., & Olson P. (2005). Mortality in over 350,000 insured Swedish dogs from 1995-2000: I. Breed-, gender-, age- and cause-specific rates. Acta Veterinaria Scandinavica, 46(3), 105-120. doi:10.1186/1751-0147-46-105
- Fleming J.M., Creevy K.E., & Promislow D.E.L. (2011). Mortality in North American dogs from 1984 to 2004: an investigation into age-, size-, and breed-related causes of death. Journal of Veterinary Internal Medicine, 25(2), 187-198. doi:10.1111/j.1939-1676.2011.0695.x
- Dobson J.M. (2013). Breed-predispositions to cancer in pedigree dogs. ISRN Veterinary Science, 2013, 941275. doi:10.1155/2013/941275
- Torres de la Riva G., Hart B.L., Farver T.B., Oberbauer A.M., Messam L.L., Willits N., & Hart L.A. (2013). Neutering dogs: effects on joint disorders and cancers in golden retrievers. PLOS ONE, 8(2), e55937. doi:10.1371/journal.pone.0055937
- Hart B.L., Hart L.A., Thigpen A.P., & Willits N.H. (2014). Long-term health effects of neutering dogs: comparison of Labrador Retrievers with Golden Retrievers. PLOS ONE, 9(7), e102241. doi:10.1371/journal.pone.0102241
- Tonomura N., Elvers I., Thomas R., Megquier K., Turner-Maier J., Howald C., et al. (2015). Genome-wide Association Study Identifies Shared Risk Loci Common to Two Malignancies in Golden Retrievers. PLOS Genetics, 11(2), e1004922. doi:10.1371/journal.pgen.1004922
- Guy M.K., Page R.L., Jensen W.A., Olson P.N., Haworth J.D., Searfoss E.E., & Brown D.E. (2015). The Golden Retriever Lifetime Study: establishing an observational cohort study with translational relevance for human health. Philosophical Transactions of the Royal Society B, 370(1673), 20140230. doi:10.1098/rstb.2014.0230
- Labadie J., Swafford B., DePena M., Tietje K., Page R., & Patterson-Kane J. (2022). Cohort profile: The Golden Retriever Lifetime Study (GRLS). PLOS ONE, 17(6), e0269425. doi:10.1371/journal.pone.0269425