Golden Retriever Muscular Dystrophy
Golden Retriever Muscular Dystrophy, or GRMD, is rare in the pet-breeding population but important for two reasons. First, it is a severe inherited disease with serious welfare consequences for affected dogs and should not be treated as a minor genetic curiosity. Second, it has unusual scientific visibility because the canine model is valuable in Duchenne muscular dystrophy research and has been maintained in research colonies precisely for that reason. Both things are true at once, and breeding interpretation has to hold them together carefully, because the same condition that has scientific value in one context has no place in pet breeding in another. Documented
What It Means
The disease at the molecular and clinical level
GRMD is the canine dystrophin-deficiency model most closely parallel to human Duchenne muscular dystrophy. It is caused by a mutation affecting the dystrophin gene, which produces a large structural protein that supports the muscle cell membrane during contraction. When dystrophin is absent or nonfunctional, the muscle cells are mechanically fragile, and ordinary use causes accumulating cellular damage that the body cannot fully repair. Over time, the affected muscle fibers degenerate and are replaced with fibrous and fatty tissue, progressively reducing muscle function across the dog's body.
Clinically, this is not a mild cosmetic problem or a manageable lifelong inconvenience. Affected dogs can show progressive muscle weakness that worsens as they grow, abnormal gait and reduced exercise tolerance, poor physical development with stunted growth, difficulty with basic activities that other dogs perform without effort, and significant welfare compromise over time as the disease advances. Documented The trajectory is not curable with current veterinary medicine, and the prognosis for affected dogs is guarded at best. This is a disease that the responsible pet-breeding community treats with the full weight its clinical impact deserves.
Why X-linked inheritance changes everything
The inheritance pattern is X-linked recessive, which makes the breeding math meaningfully different from the usual autosomal recessive examples most breeders and families are familiar with. Because males have one X chromosome and one Y chromosome, a male inheriting the affected X from his mother has no second X to provide a working copy of the dystrophin gene, and the disease expresses in that male. He does not need two copies to become affected because there is no possibility of a second X in the first place.
Females have two X chromosomes. A female with one affected X and one normal X is a carrier, and her clinical picture is usually much milder than a male's full expression because the normal copy on her other X chromosome provides functional dystrophin, though carrier females can show subtle signs in some cases due to X-inactivation effects. Carrier females can pass the affected X on to offspring: on average, half of a carrier female's sons will inherit the affected X and be fully affected, and half of her daughters will inherit it and be carriers themselves.
The practical breeding implications are stark. A carrier female bred to any male (affected or not) can produce affected males, because the sons only need one copy of the affected X to express the disease. Documented Because of this, the management of X-linked conditions requires testing females of at-risk lines, tracing the allele through pedigrees, and refusing to breed carrier females even if the carrier status itself causes them no direct harm. The usual carrier-to-clear strategy that works for autosomal recessive conditions is not adequate here, because a carrier female bred to a clear male still produces carrier daughters and affected sons.
Why GRMD matters as a teaching example
That is why GRMD matters as a genetics lesson even though it is uncommon in ordinary Golden breeding. It shows how inheritance pattern changes management in fundamental ways. The breeder is not just asking whether a dog carries the allele. The breeder is asking how sex-linked transmission changes the risk structure for sons and daughters, how carrier status in a female is biologically different from carrier status in an autosomal recessive context, and why the usual carrier-to-clear mental model does not protect against X-linked diseases the way it protects against autosomal ones.
This is also why the distinction between inheritance patterns is not academic pedantry but a practical requirement for good breeding. A breeder who only knows the autosomal-recessive carrier management framework will mishandle X-linked conditions, and the mishandling can have severe consequences when the disease in question is as devastating as GRMD. Documented Understanding the full range of Mendelian inheritance patterns is part of the basic competence responsible breeders need.
The tension between research value and breeding practice
GRMD also occupies an unusual place in science. Because it models human Duchenne pathology more closely than most other animal systems available for research, it has been maintained in dedicated research colonies where the canine disease model is genuinely valuable for studying disease mechanisms, testing potential therapies, and developing translational interventions that may eventually benefit both humans and dogs. Documented That research importance is real, and the scientific work done with these colonies has contributed to the current understanding of dystrophin-related disease.
That research context should not be confused with pet-breeding acceptability. In a companion-breeding program, the humane goal is to avoid producing affected dogs entirely, and the existence of research colonies does not change that obligation. A pet breeder has no reason to knowingly maintain the allele in a breeding line, and every reason to test for it and exclude carriers from the program if the allele is known to be present. The two contexts (research breeding and pet breeding) have different goals, different welfare considerations, and different acceptable outcomes, and conflating them would be a serious error.
GRMD today in pet populations
In the current Golden Retriever pet-breeding population, GRMD is not a common concern because the allele is not widely distributed and most breeding lines have no known history of it. Families buying a Golden Retriever puppy from a typical responsible breeder are unlikely to encounter GRMD as part of the testing discussion, and the absence of GRMD from a breeder's disclosure list is not a red flag in most cases. The page exists not because GRMD is a frequent practical concern but because understanding it teaches the broader lesson about X-linked inheritance and the distinction between research and pet breeding contexts.
Where GRMD does matter is in the minority of lines where the allele is present or suspected. In those cases, responsible management requires testing, pedigree tracing, and exclusion of carriers from the breeding program. The rarity of the condition does not reduce the obligation to handle it properly when it does appear.
Why It Matters for Your Dog
Families are unlikely to encounter GRMD often in normal pet-breeding contexts, but the page matters because it teaches three useful lessons that apply beyond this specific condition.
First, not every inherited disease in dogs is autosomal recessive. The carrier-clear-affected framework that works for most commonly tested conditions does not cover X-linked diseases the same way, and families who only know the autosomal framework may misunderstand what carrier status means in an X-linked context. A female labeled as a carrier for an X-linked condition is in a different risk category than a female labeled as a carrier for an autosomal recessive condition, and the difference matters.
Second, some rare conditions have outsized scientific importance without becoming acceptable risks for pet breeding. The existence of research interest in GRMD does not mean pet breeders should be relaxed about it. Documented The two contexts are genuinely different, and the welfare obligations of a pet breeder are not softened by the existence of research colonies elsewhere.
Third, disease rarity does not reduce the obligation to manage it properly in known-risk lines. A breeder who knows or suspects GRMD risk in a line cannot treat the rarity of the condition as an excuse for informal management. The obligation to prevent affected puppies from being produced is the same whether the disease is common or rare.
For JB, the practical interpretation is simple. GRMD belongs in the category of conditions that should be functionally eliminated from pet breeding through responsible knowledge of line risk and carrier status. The program's lines have no known history of GRMD, but the principle is firm: if the disease were ever identified in a line, the response would be immediate, complete exclusion of the affected dogs and their at-risk relatives from the breeding program, not carrier management. The severity of the disease and the lack of any realistic way to avoid affected offspring from carrier females make exclusion the only defensible approach.
What This Cannot Predict
Testing cannot identify GRMD risk in lines where the allele has not been characterized or suspected, because current panels focus on known variants rather than discovering new ones.
It cannot predict whether subtle carrier-female phenotypes will appear in specific individuals, because X-inactivation is stochastic and the clinical consequences are variable.
And it cannot resolve the broader question of what to do about research colonies, which involves ethical considerations beyond the scope of molecular testing. Documented
In pet breeding, the relevant question is simpler: if the allele is present in a line, manage it out; if it is not, standard testing protocols are adequate.
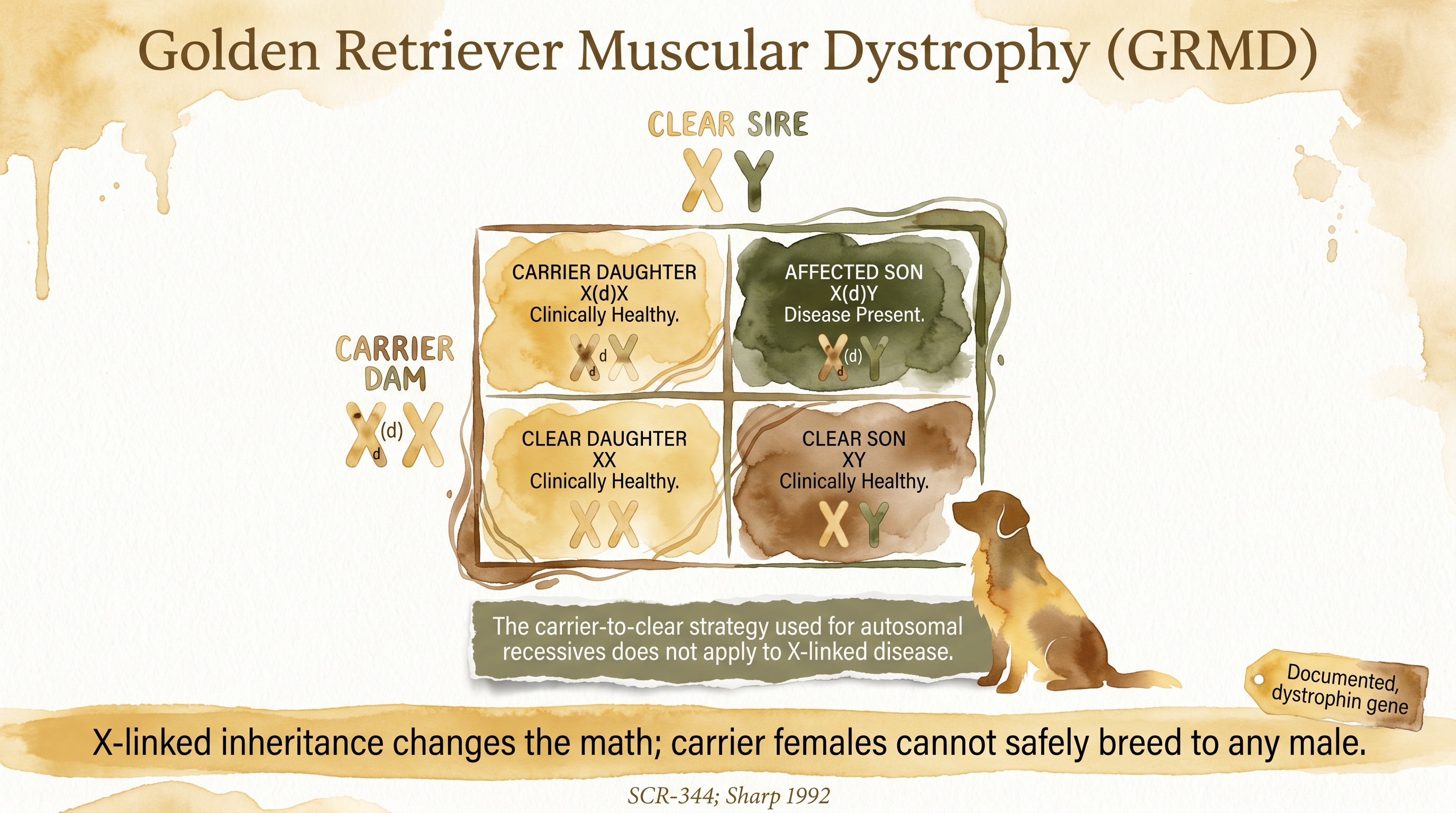
X-linked inheritance changes the math; carrier females cannot safely breed to any male.
Key Takeaways
- GRMD is a severe dystrophin-related muscular disease, not a minor inherited inconvenience.
- Its X-linked recessive inheritance changes the breeding logic compared with common autosomal recessive examples.
- The disease is scientifically important as a Duchenne model, but that research relevance does not justify its presence in pet breeding.
- In known-risk lines, responsible breeding aims to prevent affected offspring entirely.
- X-linked inheritance requires exclusion of carrier females rather than carrier-to-clear management.
The Evidence
- Golden Retriever inherited-disease literatureGolden Retrievers and dogs
Golden Retriever Muscular Dystrophy is a severe dystrophin-related myopathy with X-linked recessive inheritance and significant welfare consequences for affected males. - Comparative disease-model literaturedogs and humans
The canine GRMD model is scientifically important because it parallels important features of human Duchenne muscular dystrophy.
- Canine medical-genetics logicdogs
Known carrier-risk lines should be managed to prevent production of affected puppies, and X-linked inheritance requires special attention because male offspring are differently exposed than females. - X-linked disease management literaturemammals generally
X-linked recessive conditions cannot be managed through the same carrier-to-clear strategy used for autosomal recessives, because carrier females can produce affected male offspring regardless of the sire's status.
No published epidemiological study has surveyed the frequency of GRMD mutations in contemporary Golden Retriever breeding populations or identified which specific lines carry allelic risk.
SCR References
Sources
- Sharp N.J., Kornegay J.N., Van Camp S.D., Herbstreith M.H., Secore S.L., Kettle S., et al. (1992). An error in dystrophin mRNA processing in golden retriever muscular dystrophy, an animal homologue of Duchenne muscular dystrophy. Genomics, 13(1), 115-121. doi:10.1016/0888-7543(92)90210-J
- Kornegay J.N. (2017). The golden retriever model of Duchenne muscular dystrophy. Skeletal Muscle, 7(1), 9. doi:10.1186/s13395-017-0124-z
- Shaffer L.G., Sundin K., Geretschlaeger A., Segert J., Swinburne J.E., Royal R., Loechel R., Ramirez C.J., & Ballif B.C. (2018). Standards and guidelines for canine clinical genetic testing laboratories. Human Genetics, 138(5), 493-499. doi:10.1007/s00439-018-1954-4
- Slutsky J., Officer A., & Giger U. (2013). A web resource on DNA tests for canine and feline hereditary diseases. The Veterinary Journal, 197(2), 182-187. doi:10.1016/j.tvjl.2013.02.021
- Farrell L.L., Schoenebeck J.J., Wiener P., Clements D.N., & Summers K.M. (2015). The challenges of pedigree dog health: approaches to combating inherited disease. Canine Genetics and Epidemiology, 2, 3. doi:10.1186/s40575-015-0014-9