Transgenerational Epigenetic Effects
Compound evidence detail1 SCR / 2 parts
- Documentedthe Awalt 2024 finding that NR3C1 and OXTR methylation in dogs covaries with documented early-life experience
- Heuristicthe JB inference that those documented methylation differences in dogs predict specific later health or developmental outcomes
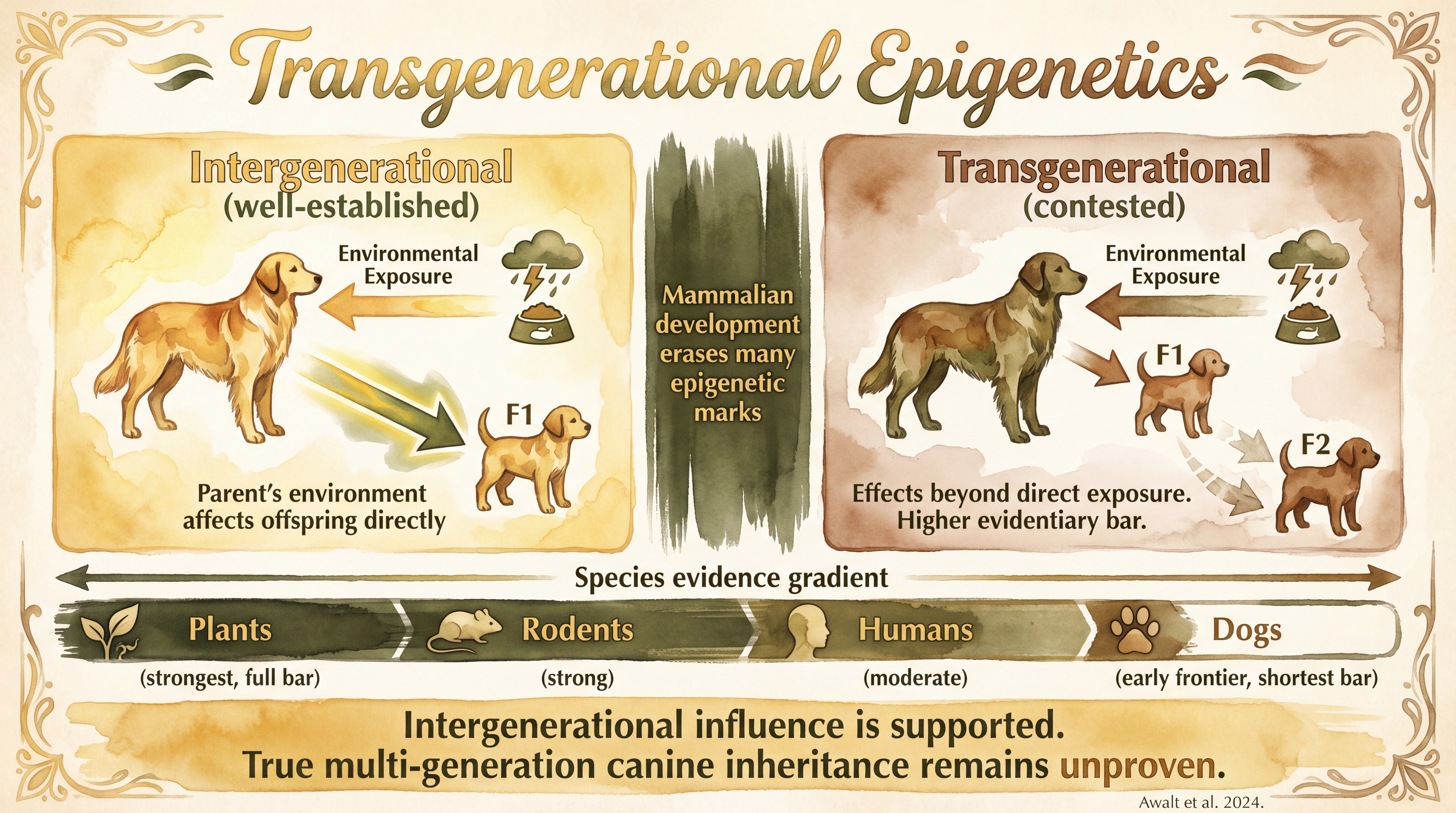
Transgenerational epigenetic effects refer to environmentally influenced molecular changes that are transmitted across generations rather than being confined to the directly exposed individual. This is one of the most discussed and most contested areas of epigenetics. The safest scientific position is that intergenerational effects are well established in many systems, while true transgenerational inheritance in mammals is more selective, harder to prove, and much less well established in dogs. Mixed Evidence
What It Means
The first thing this topic requires is a vocabulary distinction. Intergenerational effects usually mean a parent's environment affects the offspring, or F1. Observed-JB True transgenerational effects usually mean the effect persists into F2 or beyond without direct exposure being the full explanation. That distinction gets even more technical depending on whether one is talking about maternal pregnancy exposure, paternal germline pathways, or other developmental timing issues. But the basic point is simple: not every cross-generation effect is a fully transgenerational one.
Mammalian development includes extensive epigenetic reprogramming. Many marks are erased or reset as gametes and embryos develop. That makes true stable inheritance of environmentally induced marks a high evidentiary bar. To prove true transgenerational inheritance, researchers need to separate direct exposure effects, developmental uterine effects, maternal behavior effects, social transmission, and genuine germline-linked persistence. Documented That is why this topic remains methodologically difficult even when the biological intuition is strong.
Why It Matters for Your Dog
The strongest part of the literature is not the boldest-sounding part. It is the intergenerational part. It is well supported across many systems that parental environment can affect offspring development, maternal stress and care can shape offspring physiology, and early-life epigenetic marks can be associated with later stress regulation. That is already a significant biological claim. The field does not need universal transgenerational inheritance to matter.
Dogs currently add more to the intergenerational and early-life-programming story than to the true transgenerational one. SCR-094 supports direct dog evidence that early-life adversity is associated with altered methylation on stress- and bonding-related genes. That is an important canine contribution. What it does not directly show is a fully demonstrated dog F2 or F3 inheritance chain for environmentally induced epigenetic marks. Documented So the dog literature is relevant to the broader conversation, but it does not yet justify strong claims that specific canine epigenetic stress marks are known to pass stably across multiple generations.
Where the Evidence Is Strongest
The strength gradient looks roughly like this: plants show the strongest clear transgenerational cases, rodents show meaningful supportive evidence under some paradigms, humans show suggestive but heavily confounded evidence, and dogs show early and limited evidence. Estimated This does not mean the canine idea is wrong. It means the right rhetorical ceiling is lower.
Why This Still Matters for Dog Development
Even if one sets true transgenerational claims aside, the concept still matters because it keeps attention on how developmental conditions can matter before a puppy has any explicit training history. Mixed Evidence The responsible phrasing is: early-life environment matters directly, parental and maternal conditions may matter indirectly, and full multi-generation epigenetic inheritance in dogs remains a research frontier. That is strong enough to be scientifically useful without turning a difficult field into a certainty claim.
The maternal-care layer is strongest when it focuses on direct developmental programming and intergenerational influence. Stronger multi-generation inheritance claims should stay more provisional in dogs.
Parental stress experience can mark offspring gene expression through epigenetic transmission across generations.
Key Takeaways
- Intergenerational and transgenerational effects are not the same claim.
- True mammalian transgenerational epigenetic inheritance is difficult to prove because reprogramming wipes many marks.
- Dogs currently contribute more to the early-life and intergenerational story than to a fully established transgenerational one.
- The topic is scientifically useful, but it needs careful ceilings rather than bold inheritance claims.
The Evidence
This entry uses observed claim-level tags beyond the dedicated EvidenceBlocks below. These tags mark JB program observation or practice-derived claims that need dedicated EvidenceBlock coverage in a later content pass.
This entry uses estimated claim-level tags beyond the dedicated EvidenceBlocks below. These tags mark approximate ranges or timing claims that should remain bounded by the cited sources.
This entry uses mixed-evidence claim-level tags beyond the dedicated EvidenceBlocks below. These tags mark claims that combine documented findings with observed practice, heuristic application, or unresolved gaps.
- Rodent transgenerational epigenetics literaturerodents
Supports some cases of environmentally linked effects extending beyond directly exposed offspring, while remaining highly paradigm-dependent. - Human and mammalian review literaturehumans and multiple mammals
Consistently distinguishes well-supported intergenerational effects from more contested claims of true transgenerational inheritance.
- Awalt, S. L. et al. (2024)domestic dogs
Supports direct canine association between early-life adversity and epigenetic marks on stress- and bonding-related genes, but not a fully established multi-generation inheritance chain.
- SCR-094 boundarydomestic dogs
Current dog evidence supports early-life epigenetic association more strongly than it supports true transgenerational inheritance across multiple generations.
No study has measured epigenetic marks in successive generations of related dogs to determine whether environmentally induced marks on the F0 persist measurably in F1 or F2 without direct exposure.
No multispecies comparison has been conducted to estimate the likelihood of stable epigenetic inheritance across generations in dogs relative to other mammalian species.
SCR References
Sources
- Awalt, S. L., et al. (2024). A dog's life: Early life histories influence methylation of glucocorticoid (NR3C1) and oxytocin (OXTR) receptor genes, cortisol levels, and attachment styles. Developmental Psychobiology.
- Mammalian and rodent epigenetic inheritance review
- Human and mammalian review package: supports cross-species epigenetic plausibility, not direct dog transgenerational proof.
- Rodent transgenerational epigenetics package: supports mechanism plausibility only; no direct dog transgenerational inheritance claim.
- SCR-094 boundary: direct dog evidence is early-life adversity methylation association, not inherited transgenerational transmission.