Stress and Telomeres in Dogs
Compound evidence detail1 SCR / 2 parts
- Documentedthe canine telomere-shortening evidence under documented chronic stressors such as kenneling, shelter conditions, low activity, and large group housing (Dutra 2025, N=250)
- Heuristicthe JB extrapolation that chronic household excitability produces equivalent telomere shortening to documented institutional stressors and reduces canine lifespan
Telomeres are protective DNA caps at the ends of chromosomes. They shorten gradually with cell division, but that shortening can accelerate under conditions of chronic oxidative, inflammatory, and psychological strain. In humans, the stress-telomere link is well established. In dogs, direct welfare and stress-related telomere work now exists, but it is newer, narrower, and more correlational than the human literature. Documented
What It Means
What Telomeres Measure
Telomeres are often treated as a cellular aging marker. That shorthand is useful, but incomplete. Telomere length is not a single direct readout of "stress." It is shaped by age, cell turnover, oxidative burden, inflammation, disease status, and tissue and assay choice. That is why telomere pages need more caution than cortisol pages. A cortisol sample is noisy but immediate. Telomere length is slower, more cumulative, and easier to overinterpret.
Why Stress Researchers Care About Them
The appeal is simple. If chronic strain leaves a biological scar, telomeres are one candidate place to look for it.
Human stress science gave this idea much of its momentum. Epel and colleagues showed that chronic caregiving stress was associated with shorter telomeres and lower telomerase activity. Since then, the broader human literature has repeatedly linked adversity, trauma, and sustained strain with faster biological aging signatures. Documented
That cross-species background matters, but it is not enough on its own. Dog claims still need dog data.
What the Dog Studies Suggest
The canine literature is no longer empty here. Dutra and colleagues measured relative telomere length across 250 dogs in different life-history groups, including companion, shelter, police, laboratory, and recently rehomed dogs. The pattern fit the broader welfare logic: dogs in more chronically adverse or institution-like conditions tended to show shorter telomeres. Documented
The same study also reported associations with housing and social structure. Documented Dogs housed in kennels, outdoor arrangements, or laboratory environments showed shorter relative telomere length than dogs in more buffered settings. Larger group living was also associated with shorter telomeres, which the source layer interprets as consistent with chronic interpersonal or environmental load. That does not make telomeres a clean welfare verdict. It does show that direct dog research is beginning to detect cumulative-strain signals at the cellular level.
Weixlbraun and colleagues added a second, more behavior-linked finding. In aging pet dogs, trainability emerged as the strongest behavioral correlate of preserved telomere length. This is an intriguing result, but it is still correlational. It does not prove that structured learning causes longer telomeres. It shows that behavior profile and cellular-aging markers can travel together in dogs. Documented
Why It Matters for Your Dog
What the Evidence Does Not Yet Support
This is where slippage risk gets real. The dog evidence does not yet prove that ordinary household excitement, lively greetings, or generic overstimulation in everyday family life measurably shortens telomeres. The strongest canine findings come from more substantial chronic-load contexts such as sheltering, institutional housing, kennel living, and broader environmental adversity.
That stress-type boundary matters because it is easy to jump from chronic adverse conditions being associated with shorter canine telomeres to any excitable household aging the dog at the cellular level. The first claim is defensible. The second is not yet directly demonstrated.
How This Fits the Allostatic-Load Picture
Telomeres are best understood here as one possible cumulative-outcome marker, not as a replacement for the rest of stress physiology. If cortisol, autonomic strain, immune disruption, and inflammatory load are the active systems doing the work of adaptation, telomeres may be one place where the long-term cost becomes visible. That is why telomere research belongs next to allostatic load and chronic-stress pages, not in isolation.
At the same time, the welfare-biomarker literature remains cautious. Recent reviews note real construct-validity problems, including differences between tissues, assay methods, and what exactly any one telomere measure is capturing.
Why the Page Is Marked Mixed
The evidence is mixed not because the biology is implausible, but because the layers are uneven. Ambiguous Human evidence for stress-related telomere attrition is strong, direct dog evidence now exists, the dog literature is still early and largely correlational, and the jump from documented adverse contexts to ordinary household arousal remains a gap. That makes this a strong concept with a narrower canine application than casual summaries often imply.
The calmness layer sometimes treats cumulative stress as something that can leave lasting biological residue. This page supports that general direction while keeping the narrower scientific boundary intact: canine telomere findings currently document cumulative-strain associations more clearly than they document any specific everyday household-arousal effect.
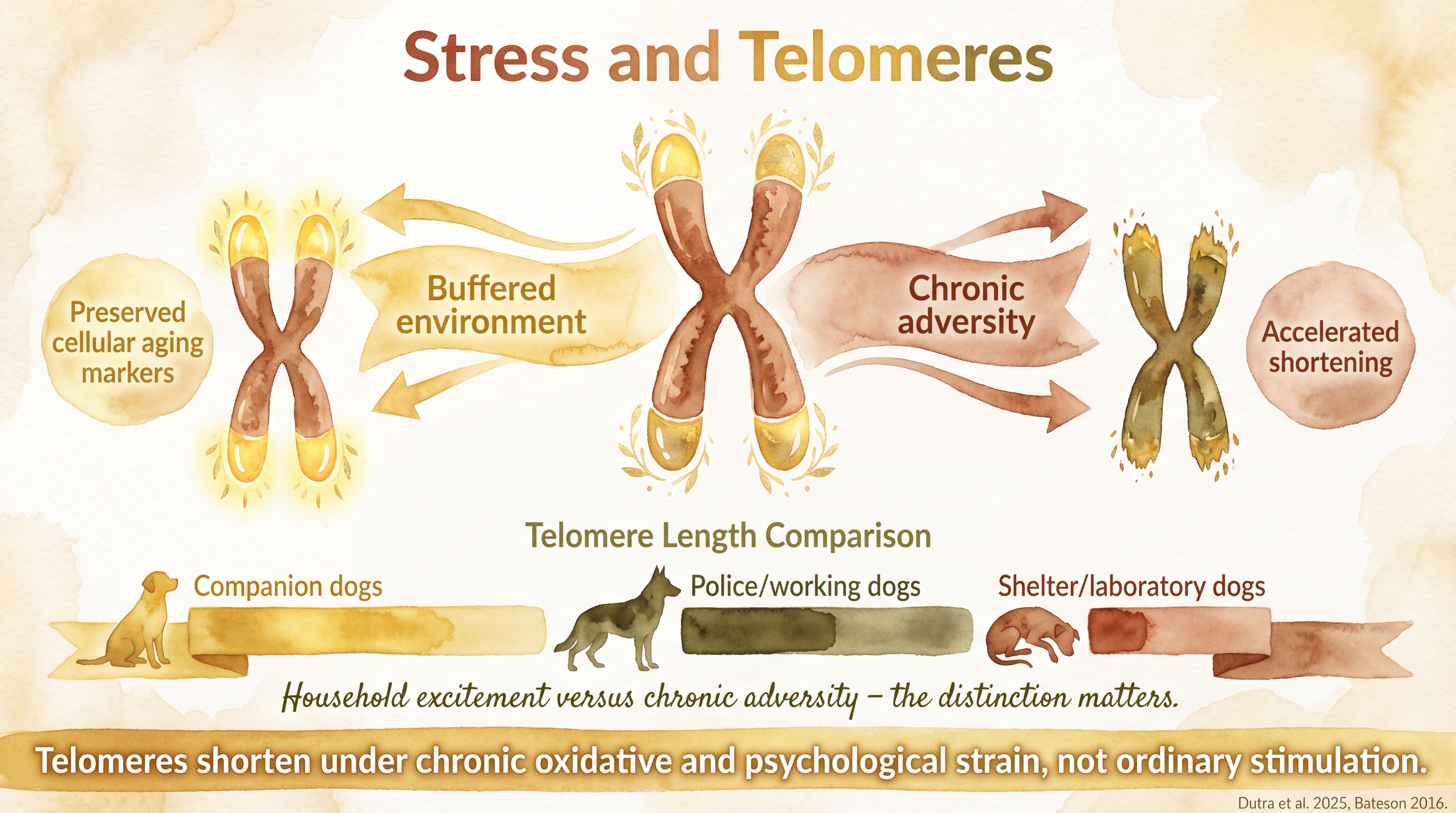
Chronic stress accelerates telomere shortening - providing a biological clock that records cumulative environmental burden.
Key Takeaways
- Telomeres are cumulative cellular-aging markers, not simple one-number stress meters.
- Human evidence linking chronic stress and shorter telomeres is strong.
- Dogs now have direct telomere studies consistent with the same broad pattern, but the literature is still early and mostly correlational.
- The strongest canine evidence comes from chronic adverse environments, so ordinary household-arousal claims still need restraint.
The Evidence
This entry uses ambiguous claim-level tags beyond the dedicated EvidenceBlocks below. These tags mark claims where the literature remains unsettled or multiple interpretations coexist.
- Epel, E. S. et al. (2004)humans
Chronic caregiving stress was associated with shorter telomeres and lower telomerase activity. - Bateson, M. (2016)multiple species
Reviewed telomere attrition as a cumulative-stress and welfare marker while emphasizing interpretive caution.
- Dutra, L. M. L. et al. (2025)domestic dogs
Measured relative telomere length across 250 dogs and linked shorter telomeres with more adverse or institutional living contexts. - Weixlbraun, J. et al. (2025)domestic dogs
Found trainability to be the strongest behavioral correlate of preserved telomere length in aging pet dogs, while remaining correlational rather than causal. - Fick, L. J. et al. (2012)domestic dogs
Showed that telomere biology is a meaningful aging variable in dogs, even though this was not itself a welfare-stress design.
- SCR-015 boundarydomestic dogs
Direct dog evidence now exists for stress-related telomere shortening, but the strongest canine evidence comes from chronic adverse environments rather than from ordinary household excitability. - Cobb, M. L. et al. (2025)domestic dogs
Reviewed welfare biomarkers and cautioned against overinterpreting any single physiological marker as a complete stress verdict.
No study has measured whether improved housing or reduced stress in dogs results in measurable telomere lengthening or stabilization, leaving unclear whether telomere shortening is reversible.
No longitudinal study has tracked the same dogs' telomere length from puppyhood through old age to establish a normal developmental trajectory and to measure how much variation is expected from genetics alone.
SCR References
Sources
- Bateson, M. (2016). Cumulative stress in research animals: Telomere attrition as a biomarker in a welfare context. BioEssays, 38(2), 201-212.
- Cobb, M. L., Jimenez, A. G., & Dreschel, N. A. (2025). Beyond cortisol! Physiological indicators of welfare for dogs. Journal of Applied Animal Welfare Science.
- Dutra, L. M. L., et al. (2025). Telomere Tales: Exploring the impact of stress, sociality, and exercise on dogs' cellular aging. Veterinary Sciences.
- Epel, E. S., et al. (2004). Accelerated telomere shortening in response to life stress. Proceedings of the National Academy of Sciences, 101(49), 17312-17315.
- Fick, L. J., et al. (2012). Telomere length correlates with life span of dog breeds. Cell Reports, 2(6), 1530-1536.
- Weixlbraun, J., et al. (2025). Impact of trainability on telomere dynamics of pet dogs. PLOS ONE.